Theracodophen-325
Theracodophen-325
FULL PRESCRIBING INFORMATION
DESCRIPTION
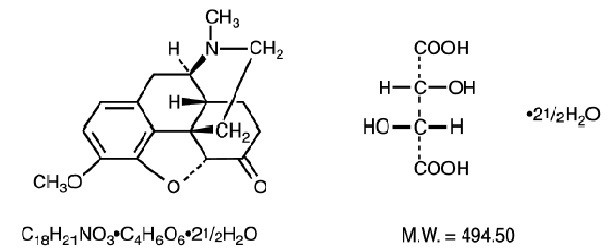
Hydrocodone Bitartrate and Acetaminophen Tablets, USP are supplied in tablet form for oral administration. Hydrocodone bitartrate is an opioid analgesic and antitussive and occurs as fine, white crystals or as a crystalline powder. It is affected by light. The chemical name is 4,5α-Epoxy-3-methoxy-17-methylmorphinan-6one tartrate (1:1) hydrate (2:5). It has the following structural formula:
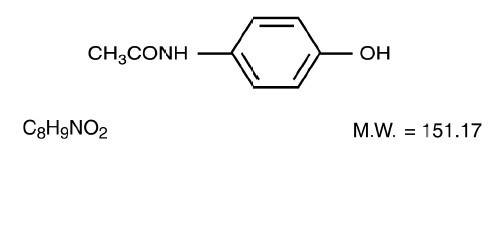
Acetaminophen, 4' -Hydroxyacetanilide, a slightly bitter, white, odorless, crystalline powder, is a non-opiate, non-salicylate analgesic and antipyretic, It has the following structural formula:
Each Hydrocodone Bitartrate and Acetaminophen Tablets, USP contains:
Hydrocodone Bitartrate, USP. . . . . . . . . 10 mg
Acetaminophen, USP. . . . . . . . . . . . . 325 mg
In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose, povidone, pregelatinized starch and stearic acid.
Meets USP Dissolution Test 1.
CLINICAL PHARMACOLOGY
Hydrocodone is a semisynthetic opioid analgesic and antitussive with multiple actions qualitatively similar to those of codeine. Most of these involve the central nervous system and smooth muscle. The precise mechanism of action of hydrocodone and other opiates is not known, although it is believed to relate to the existence of opiate receptors in the central nervous system. In addition to analgesia, opioids may produce drowsiness, changes in mood and mental clouding.
The analgesic action of acetaminophen involves peripheral influences, but the specific mechanism is as yet undetermined. Antipyretic activity is mediated through hypothalamic heat regulating centers. Acetaminophen inhibits prostaglandin synthetase. Therapeutic doses of acetaminophen have negligible effects on the cardiovascular or respiratory systems: however, toxic doses may cause circulatory failure and rapidm shallow breathing,
:
Uses
INDICATIONS AND USAGE
Hydrocodone Bitartrate and Acetaminophen Tablets, USP are indicated for the relief of moderate to moderately severe pain.
CONTRAINDICATIONS
Hydrocodone bitartrate and acetaminophen tablets, USP should not be administered to patients who have previously exhibited hypersensitivity to hydrocodone or acetaminophen, or any other component of this product.
Patients known to be hypersensitive to other opioids may exhibit cross-sensitivity to hydrocodone.
WARNINGS
Respiratory Depression
At high doses or in sensitive patients, hydrocodone may produce dose-related respiratory depression by acting directly on the brain stem respiratory center. Hydrocodone also affects the center that controls respiratory rhythm, and may produce irregular and periodic breathing.
Head Injury and Increased Intracranial Pressure
The respiratory depressant effects of opioids and their capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or a pre-existing increase in intracranial pressure. Furthermore, opioids produce adverse reactions which may obscure the clinical course of patients with head injuries.
Acute Abdominal Conditions
The administration of opioids may obscure the diagnosis or clinical course of patients with acute abdominal conditions.
PRECAUTIONS
General
Special Risk Patients: As with any opioid analgesic agent, hydrocodone bitartrate and acetaminophen tablets should be used with caution in elderly or debilitated patients and those with severe impairment of hepatic or renal function, hypothyroidism, Addison's disease, prostatic hypertrophy or urethral stricture. The usual precautions should be observed and the possibility of respiratory depression should be kept in mind.
Cough Reflex: Hydrocodone suppresses the cough reflex; as with all opioids, caution should be exercised when hydrocodone bitartrate and acetaminophen tablets are used postoperatively and in patients with pulmonary disease.
Information for Patients
Hydrocodone, like all opioids, may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery; patients should be cautioned accordingly.
Alcohol and other CNS depressants may produce an additive CNS depression, when taken with this combination product, and should be avoided.
Hydrocodone may be habit-forming. Patients should take the drug only for as long as it is prescribed, in the amounts prescribed, and no more frequently than prescribed.
Drug Interactions
Patients receiving other opioids, antihistamines, antipsychotics, anti-anxiety agents, or other CNS depressants (including alcohol) concomitantly with hydrocodone bitartrate and acetaminophen tablets may exhibit an additive CNS depression. When combined therapy is contemplated, the dose of one or both agents should be reduced.
The use of MAO inhibitors or tricyclic antidepressants with hydrocodone preparations may increase the effect of either the antidepressant or hydrocodone.
Drug/Laboratory Test Interactions
Acetaminophen may produce false-positive test results for urinary 5-hydroxyindoleacetic acid.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No adequate studies have been conducted in animals to determine whether hydrocodone or acetaminophen have a potential for carcinogenesis, mutagenesis, or impairment of fertility.
Pregnancy
Teratogenic Effects: Pregnancy Category C: There are no adequate and well-controlled studies in pregnant women. Hydrocodone bitartrate and acetaminophen tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects: Babies born to mothers who have been taking opioids regularly prior to delivery will be physically dependent. The withdrawal signs include irritability and excessive crying, tremors, hyperactive reflexes, increased respiratory rate, increased stools, sneezing, yawning, vomiting, and fever. The intensity of the syndrome does not always correlate with the duration of maternal opioid use or dose. There is no consensus on the best method of managing withdrawal.
Labor and Delivery
As with all opioids, administration of this product to the mother shortly before delivery may result in some degree of respiratory depression in the newborn, especially if higher doses are used.
Nursing Mothers
Acetaminophen is excreted in breast milk in small amounts, but the significance of its effects on nursing infants is not known. It is not known whether hydrocodone is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from hydrocodone and acetaminophen, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatric Use
Clinical studies of hydrocodone bitartrate and acetaminophen did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Hydrocodone and the major metabolites of acetaminophen are known to be substantially excreted by the kidney. Thus the risk of toxic reactions may be greater in patients with impaired renal function due to the accumulation of the parent compound and/or metabolites in the plasma. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Hydrocodone may cause confusion and over-sedation in the elderly; elderly patients generally should be started on low doses of hydrocodone bitartrate and acetaminophen tablets and observed closely.
ADVERSE REACTIONS
The most frequently reported adverse reactions are light-headedness, dizziness, sedation, nausea and vomiting. These effects seem to be more prominent in ambulatory than in non-ambulatory patients, and some of these adverse reactions may be alleviated if the patient lies down.
Other adverse reactions include:
Central Nervous System: Drowsiness, mental clouding, lethargy, impairment of mental and physical performance, anxiety, fear, dysphoria, psychic dependence, mood changes.
Gastrointestinal System: Prolonged administration of hydrocodone bitartrate and acetaminophen tablets may produce constipation.
Genitourinary System: Ureteral spasm, spasm of vesical sphincters and urinary retention have been reported with opiates.
Respiratory Depression: Hydrocodone bitartrate may produce dose-related respiratory depression by acting directly on the brain stem respiratory centers (see OVERDOSAGE ).
Special Senses: Cases of hearing impairment or permanent loss have been reported predominantly in patients with chronic overdose.
Dermatological: Skin rash, pruritus.
The following adverse drug events may be borne in mind as potential effects of acetaminophen: allergic reactions, rash, thrombocytopenia, agranulocytosis. Potential effects of high dosage are listed in the OVERDOSAGE section.
DRUG ABUSE AND DEPENDENCE
Controlled Substance: Hydrocodone bitartrate and acetaminophen tablets are classified as a Schedule III controlled substance.
Abuse and Dependence: Psychic dependence, physical dependence, and tolerance may develop upon repeated administration of opioids; therefore, this product should be prescribed and administered with caution. However, psychic dependence is unlikely to develop when hydrocodone bitartrate and acetaminophen tablets are used for a short time for the treatment of pain.
Physical dependence, the condition in which continued administration of the drug is required to prevent the appearance of a withdrawal syndrome, assumes clinically significant proportions only after several weeks of continued opioid use, although some mild degree of physical dependence may develop after a few days of opioid therapy. Tolerance, in which increasingly large doses are required in order to produce the same degree of analgesia, is manifested initially by a shortened duration of analgesic effect, and subsequently by decreases in the intensity of analgesia. The rate of development of tolerance varies among patients.
OVERDOSAGE
Following an acute overdosage, toxicity may result from hydrocodone or acetaminophen.
Signs and Symptoms
Hydrocodone: Serious overdose with hydrocodone is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, and sometimes bradycardia and hypotension. In severe overdosage, apnea, circulatory collapse, cardiac arrest and death may occur.
Acetaminophen: In acetaminophen overdosage: dose-dependent, potentially fatal hepatic necrosis is the most serious adverse effect. Renal tubular necrosis, hypoglycemic coma, and thrombocytopenia may also occur.
Early symptoms following a potentially hepatotoxic overdose may include: nausea, vomiting, diaphoresis and general malaise. Clinical and laboratory evidence of hepatic toxicity may not be apparent until 48 to 72 hours post-ingestion.
In adults, hepatic toxicity has rarely been reported with acute overdoses of less than 10 grams, or fatalities with less than 15 grams.
Treatment
A single or multiple overdose with hydrocodone and acetaminophen is a potentially lethal polydrug overdose, and consultation with a regional poison control center is recommended.
Immediate treatment includes support of cardiorespiratory function and measures to reduce drug absorption. Vomiting should be induced mechanically, or with syrup of ipecac, if the patient is alert (adequate pharyngeal and laryngeal reflexes). Oral activated charcoal (1 g/kg) should follow gastric emptying. The first dose should be accompanied by an appropriate cathartic. If repeated doses are used, the cathartic might be included with alternate doses as required. Hypotension is usually hypovolemic and should respond to fluids. Vasopressors and other supportive measures should be employed as indicated. A cuffed endotracheal tube should be inserted before gastric lavage of the unconscious patient and, when necessary, to provide assisted respiration.
Meticulous attention should be given to maintaining adequate pulmonary ventilation. In severe cases of intoxication, peritoneal dialysis, or preferably hemodialysis may be considered. If hypoprothrombinemia occurs due to acetaminophen overdose, vitamin K should be administered intravenously.
Naloxone, an opioid antagonist, can reverse respiratory depression and coma associated with opioid overdose. Naloxone hydrochloride 0.4 mg to 2 mg is given parenterally. Since the duration of action of hydrocodone may exceed that of the naloxone, the patient should be kept under continuous surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. An opioid antagonist should not be administered in the absence of clinically significant respiratory or cardiovascular depression.
If the dose of acetaminophen may have exceeded 140 mg/kg, acetylcysteine should be administered as early as possible. Serum acetaminophen levels should be obtained, since levels four or more hours following ingestion help predict acetaminophen toxicity. Do not await acetaminophen assay results before initiating treatment. Hepatic enzymes should be obtained initially, and repeated at 24-hour intervals.
Methemoglobinemia over 30% should be treated with methylene blue by slow intravenous administration.
The toxic dose for adults for acetaminophen is 10 g.
DOSAGE AND ADMINISTRATION
Dosage should be adjusted according to the severity of the pain and the response of the patient. However, it should be kept in mind that tolerance to hydrocodone can develop with continued use and that the incidence of untoward effects is dose related.
The usual adult dosage is one or two tablets every four to six hours as needed for pain. The total daily dose should not exceed 6 tablets.
HOW SUPPLIED
Hydrocodone Bitartrate and Acetaminophen Tablets, USP, 10 mg / 325 mg are available as white to off white, scored, oblong biconvex tablets, debossed “IP 110” on obverse and bisected on the reverse. Each tablet contains 10 mg hydrocodone bitartrate and 325 mg acetaminophen.
They are supplied as follows:
Bottles of 100: NDC 53746-110-01
Bottles of 500: NDC 53746-110-05
Bottles of 1000: NDC 53746-110-10
Storage: Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F). [see USP Controlled Room Temperature].
Dispense in a tight, light-resistant container with a child-resistant closure.
Manufactured by:
Amneal Pharmaceuticals of NY
Hauppauge, NY 11788
Distributed by:
Amneal Pharmaceuticals
Glasgow, KY 42141
Rev. 12-2009
Packaged by Bryant Ranch North Hollywood CA 91605
Hydrocodone/Apad 10/325 mg (CIII)
Compare To
Norco 10/325 mg (CIII) Tablet
Wilming xx xxx Inc Pharmaceutical Group
#60 EXP 11/09
NDC 6362929476
NANA
LOT 10040 Rx
May Cause Drowsiness/No Alcohol
Take with food
PRODUCT DESCRIPTION
Primary Ingredients Theramine consists of a proprietary blend of amino acids, cocoa, caffeine, cinnamon, and flavonoids in specific proportions. These ingredients fall into the category of Generally Regarded as Safe” (GRAS) as defined by the Food and Drug Administration (FDA) (Sections 201(s) and 409 of the Federal Food, Drug, and Cosmetic Act). A GRAS substance is distinguished from a food additive on the basis of the common knowledge about the safety of the substance for its intended use. The standard for an ingredient to achieve GRAS status requires not only technical demonstration of non-toxicity and safety, but also general recognition of safety through widespread usage and agreement of that safety by experts in the field. Many ingredients have been determined by the U.S. Food and Drug Administration (FDA) to be GRAS, and are listed as such by regulation, in Volume 21 Code of Federal Regulations (CFR) Sections 182, 184, and 186.
Amino Acids
Amino Acids are the building blocks of protein. All amino acids are GRAS listed as they have been ingested by humans for thousands of years. The doses of the amino acids in Theramine are equivalent to those found in the usual human diet. Patients with pain disorders may require an increased amount of certain amino acids that cannot be obtained from normal diet alone. Tryptophan, for example, is an obligatory amino acid. The body cannot make tryptophan and must obtain tryptophan from the diet. Tryptophan is needed to produce serotonin. Serotonin is required to reduce pain. Patients with pain disorders and inflammatory conditions have altered serotonin metabolism. Some patients with pain disorders and inflammatory conditions have a resistance to the use of tryptophan that is similar to the mechanism found in insulin resistance. Patients with pain disorders and inflammatory conditions cannot acquire sufficient tryptophan from the diet to alter the perception of pain and the inflammatory process without ingesting a prohibitively large amount of calories, particularly calories from protein.
Flavonoids
Flavonoids are a group of phytochemical compounds found in all vascular plants including fruits and vegetables. They are a part of a larger class of compounds known as polyphenols. Many of the therapeutic or health benefits of colored fruits and vegetables, cocoa, red wine, and green tea are directly related to their flavonoid content. The specially formulated flavonoids found in Theramine cannot be obtained from conventional foods in the necessary proportions to elicit a therapeutic response.
Other Ingredients
Theramine contains the following inactive or other ingredients, as fillers, excipients, and colorings: magnesium stearate, microcrystalline cellulose, Maltodextrin NF, gelatin (as the capsule material).
Physical Description
Theramine is a yellow to light brown powder. Theramine contains L-Glutamine, L-Arginine, L-Histidine, and L-Serine, 5-Hydroxytryptophan as Griffonia Seed Extract, GABA, Choline Bitartrate, Cinnamon, Cocoa, Hydrolyzed Whey Protein, and Grape Seed Extract.
CLINICAL PHARMACOLOGY
Mechanism of Action
Theramine acts by restoring and maintaining the balance of the neurotransmitters; GABA, nitric oxide, serotonin, and acetylcholine that are associated with pain disorders and inflammatory conditions. Theramine stimulates the production ACTH to reduce inflammation.
Metabolism
The amino acids in Theramine are primarily absorbed by the stomach and small intestines. All cells metabolize the amino acids in Theramine. Circulating tryptophan, arginine and choline blood levels determine the production of serotonin, nitric oxide, and acetylcholine.
Excretion
Theramine is not an inhibitor of cytochrome P450 1A2, 2C9, 2C19, 2D6, or 3A4. These isoenzymes are principally responsible for 95% of all detoxification of drugs, with CYP3A4 being responsible for detoxification of roughly 50% of drugs. Amino acids do not appear to have an effect on drug metabolizing enzymes.
Uses
INDICATIONS FOR USE
Theramine is intended for the clinical dietary management of the metabolic processes of pain disorders and inflammatory conditions.
PRECAUTIONS AND CONTRAINDICATIONS
Theramine is contraindicated in an extremely small number of patients with hypersensitivity to any of the nutritional components of Theramine.
ADVERSE REACTIONS
Oral supplementation with L-tryptophan, L-arginine or choline at high doses up to 15 grams daily is generally well tolerated. The most common adverse reactions of higher doses — from 15 to 30 grams daily — are nausea, abdominal cramps, and diarrhea. Some patients may experience these symptoms at lower doses. The total combined amount of amino acids in each Theramine capsule does not exceed 400 mg.
DRUG INTERACTIONS
Theramine does not directly influence the pharmacokinetics of prescription drugs. Clinical experience has shown that administration of Theramine may allow for lowering the dose of co-administered drugs under physician supervision.
OVERDOSE
There is a negligible risk of overdose with Theramine as the total dosage of amino acids in a one month supply (90 capsules) is less than 36 grams. Overdose symptoms may include diarrhea, weakness, and nausea.
POST-MARKETING SURVEILLANCE
Post-marketing surveillance has shown no serious adverse reactions. Reported cases of mild rash and itching may have been associated with allergies to Theramine flavonoid ingredients, including cinnamon, cocoa, and chocolate. These reactions were transient in nature and subsided within 24 hours.
DOSAGE AND ADMINISTRATION
Recommended Administration For the dietary management of the metabolic processes associated with pain disorders and inflammatory conditions. Take (2) capsules one to three times daily or as directed by physician. As with most amino acid formulations Theramine should be taken without food to increase the absorption of key ingredients.
How Supplied
Theramine is supplied in purple and white, size 0 capsules in bottles of 60 or 90 capsules.
Physician Supervision
Theramine is a Medical Food product available by prescription only and must be used while the patient is under ongoing physician supervision.
U.S. patent pending.
Manufactured by Arizona Nutritional Supplements, Inc. Chandler AZ 85225
Distributed by Physician Therapeutics LLC, Los Angeles, CA 90077. www.ptlcentral.com
Copyright 2003-2006, Physician Therapeutics LLC, all rights reserved
NDC: 68405-1008-02
NDC: 68405-1008-03
Storage
Store at room temperature, 59-86OF (15-30OC) Protect from light and moisture. Theramine is supplied to physicians in a recyclable plastic bottle with a child-resistant cap.
A Convenience Packed Medical Food and Drug
Theracodophen-325
PHYSICIAN THERAPEUTICS
Theramine 90 Capsules
Hydrocodone 10mg + Acetaminophen 325mg 60 Tablets
No Refills Without Physician Authorization
Rx Only
NDC# 68405-8098-36 of this co-pack
For the Dietary Management of Pain and Inflammation.
Two capsules twice daily or as directed by physician. See product label and insert
Theramine
Medical Food
As prescribed by physician. See product label and product information insert.
Hydrocodone 10mg + Acetaminophen 325mg
Rx Drug



Theracodophen-325HYDROCODONE BITARTRATE, ACETAMINOPHEN, .GAMMA.-AMINOBUTYRIC ACID KIT
| ||||||||||||||||||||||||||||||||||||||||