Galantamine Hydrobromide
HIGHLIGHTS OF PRESCRIBING INFORMATIONHIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets USP safely and effectively. See full prescribing information for Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets USP . Galantamine Hydrobromide Extended-Release Capsules, for oral use Galantamine Tablets USP, for oral use Initial U.S. Approval: 2001INDICATIONS AND USAGEGalantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets USP are cholinesterase inhibitors indicated for the treatment of mild to moderate dementia of the Alzheimer’s type (1)DOSAGE AND ADMINISTRATION Recommended starting dosage for Galantamine Hydrobromide Extended-Release Capsules is 8 mg/day in the morning; increase dose to an initial maintenance dose of 16 mg/day after a minimum of 4 weeks. Based upon assessment of clinical benefit and tolerability, dosage may be increased to 24 mg/day after a minimum of 4 weeks at 16 mg/day. (2.1) Conversion from Galantamine Tablets to Galantamine Hydrobromide Extended-Release Capsules should occur at the same daily dosage with the last dose of Galantamine Tablets taken in the evening and starting Galantamine Hydrobromide Extended-Release Capsules once daily treatment the next morning. (2.1) Recommended starting dosage for Galantamine Tablets is 4 mg twice daily; increase dose to initial maintenance dosage of 8 mg twice daily after a minimum of 4 weeks. Based upon assessment of clinical benefit and tolerability, dosage may be increased to 12 mg twice daily after a minimum of 4 weeks at 8 mg twice daily. (2.2) Take Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets with meals. Ensure adequate fluid intake during treatment (2.2) DOSAGE FORMS AND STRENGTHS Extended-Release Capsules – 8 mg, 16 mg, 24 mg (3) Tablets – 4 mg, 8 mg, 12 mg (3) CONTRAINDICATIONSKnown hypersensitivity to galantamine hydrobromide or any excipients (4)WARNINGS AND PRECAUTIONS All patients should be considered at risk for adverse effects on cardiac conduction, including bradycardia and AV block, due to vagotonic effects on sinoatrial and atrioventricular nodes (5.2) Active or occult gastrointestinal bleeding: monitor, especially those with an increased risk for developing ulcers (5.3) Cholinomimetics may cause bladder outflow obstruction (5.4) Cholinesterase inhibitors are believed to have some potential to cause generalized convulsions. Patients with Alzheimer’s disease should be monitored closely for seizures while taking galantamine. (5.5) Monitor for respiratory adverse events in patients with a history of severe asthma or obstructive pulmonary disease (5.6) Side EffectsThe most common adverse reactions (≥ 5%) were nausea, vomiting, diarrhea, dizziness, headache, decreased appetite, and weight decreased (6.1) To report SUSPECTED ADVERSE REACTIONS, contact TEVA USA, PHARMACOVIGILANCE at 1-866-832-8537 or drug.safety@tevapharm.com; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch DRUG INTERACTIONS Potential to interfere with the activity of anticholinergic medications (7.1) Synergistic effect expected when given concurrently with succinylcholine, other cholinesterase inhibitors, similar neuromuscular blocking agents, or cholinergic agonists (7.2) USE IN SPECIFIC POPULATIONS Pregnancy: Based on animal data may cause fetal harm. (8.1)
FULL PRESCRIBING INFORMATION: CONTENTS*
- 1 GALANTAMINE HYDROBROMIDE INDICATIONS AND USAGE
- 2 GALANTAMINE HYDROBROMIDE DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 GALANTAMINE HYDROBROMIDE CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
- 6 GALANTAMINE HYDROBROMIDE ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
- 8 USE IN SPECIFIC POPULATIONS
- 10 OVERDOSAGE
- 11 GALANTAMINE HYDROBROMIDE DESCRIPTION
- 12 CLINICAL PHARMACOLOGY
- 13 NONCLINICAL TOXICOLOGY
- 14 CLINICAL STUDIES
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets USP are indicated for the treatment of mild to moderate dementia of the Alzheimer’s type.
2 DOSAGE AND ADMINISTRATION
2.1 Galantamine Hydrobromide Extended-Release Capsules
Galantamine Hydrobromide Extended-Release Capsules should be administered once daily in the morning, preferably with food.
The recommended starting dosage of Galantamine Hydrobromide Extended-Release Capsules is 8 mg/day. The dosage should be increased to the initial maintenance dose of 16 mg/day after a minimum of 4 weeks. A further increase to 24 mg/day should be attempted after a minimum of 4 weeks at 16 mg/day. Dosage increases should be based upon assessment of clinical benefit and tolerability of the previous dose.
The dosage of Galantamine Hydrobromide Extended-Release Capsules shown to be effective in a controlled clinical trial is 16 to 24 mg/day.
Patients currently being treated with Galantamine Tablets can convert to Galantamine Hydrobromide Extended-Release Capsules by taking their last dose of Galantamine Tablets in the evening and starting Galantamine Hydrobromide Extended-Release Capsules once daily treatment the next morning. Converting from Galantamine Tablets to Galantamine Hydrobromide Extended-Release Capsules should occur at the same total daily dosage.
2.2 Galantamine Immediate-Release Tablets
The dosage of Galantamine Tablets shown to be effective in controlled clinical trials is 16 to 32 mg/day given as twice daily dosing. As the dosage of 32 mg/day is less well tolerated than lower dosages and does not provide increased effectiveness, the recommended dosage range is 16 to 24 mg/day given twice daily. The dosage of 24 mg/day did not provide a statistically significant greater clinical benefit than 16 mg/day. It is possible, however, that a daily dosage of 24 mg of Galantamine Tablets might provide additional benefit for some patients.
The recommended starting dosage of Galantamine Tablets is 4 mg twice a day (8 mg/day). The dosage should be increased to the initial maintenance dosage of 8 mg twice a day (16 mg/day) after a minimum of 4 weeks. A further increase to 12 mg twice a day (24 mg/day) should be attempted after a minimum of 4 weeks at 8 mg twice a day (16 mg/day).
Dosage increases should be based upon assessment of clinical benefit and tolerability of the previous dose.
Galantamine Tablets should be administered twice a day, preferably with morning and evening meals.
Patients and caregivers should be advised to ensure adequate fluid intake during treatment. If therapy has been interrupted for more than three days, the patient should be restarted at the lowest dosage and the dosage escalated to the current dose.
The abrupt withdrawal of Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets in those patients who had been receiving dosages in the effective range was not associated with an increased frequency of adverse events in comparison with those continuing to receive the same dosages of that drug. The beneficial effects of Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets are lost, however, when the drug is discontinued.
2.3 Hepatic Impairment
Galantamine plasma concentrations may be increased in patients with moderate to severe hepatic impairment. In patients with moderately impaired hepatic function (Child-Pugh score of 7 to 9), the total daily dosage should generally not exceed 16 mg/day. The use of Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets in patients with severe hepatic impairment (Child-Pugh score of 10 to 15) is not recommended.
2.4 Renal Impairment
For patients with moderate renal impairment the dosage should generally not exceed 16 mg/day. In patients with severe renal impairment (creatinine clearance < 9 mL/min), the use of Galantamine Hydrobromide Extended-Release Capsules and Galantamine Tablets is not recommended.
3 DOSAGE FORMS AND STRENGTHS
Galantamine Hydrobromide Extended-Release Capsules contain white to off-white pellets and are available in the following strengths:
8 mg: Light beige opaque hard gelatin capsules filled with white to off-white pellets, imprinted barr over 8 mg and 1020 in black ink.
16 mg: Pink opaque hard gelatin capsules filled with white to off-white pellets, imprinted barr over 16 mg and 1021 in black ink.
24 mg: Tan opaque hard gelatin capsules filled with white to off-white pellets, imprinted barr over 24 mg and 1022 in black ink.
Galantamine Tablets are available in the following strengths:
4 mg: Yellow, round, film-coated, biconvex, unscored tablets, debossed with “stylized b” 4 on one side and 138 on the other side.
8 mg: Dark pink, round, film-coated, biconvex, unscored tablets, debossed with “stylized b” 8 on one side and 139 on the other side.
12 mg: Red, round, film-coated, biconvex, unscored tablets, debossed with “stylized b” 12 on one side and 140 on the other side.
4 CONTRAINDICATIONS
Galantamine hydrobromide extended-release capsules and galantamine tablets are contraindicated in patients with known hypersensitivity to galantamine hydrobromide or to any excipients used in the formulation.
5 WARNINGS AND PRECAUTIONS
5.1 Anesthesia
Galantamine, as a cholinesterase inhibitor, is likely to exaggerate the neuromuscular blocking effects of succinylcholine-type and similar neuromuscular blocking agents during anesthesia.
5.2 Cardiovascular Conditions
Because of their pharmacological action, cholinesterase inhibitors have vagotonic effects on the sinoatrial and atrioventricular nodes, leading to bradycardia and AV block. Bradycardia and all types of heart block have been reported in patients both with and without known underlying cardiac conduction abnormalities. Therefore, all patients should be considered at risk for adverse effects on cardiac conduction.
Patients treated with galantamine up to 24 mg/day using the recommended dosing schedule showed a dose-related increase in risk of syncope (placebo 0.7% [2/286]; 4 mg BID 0.4% [3/692]; 8 mg BID 1.3% [7/552]; 12 mg BID 2.2% [6/273]).
5.3 Gastrointestinal Conditions
Through their primary action, cholinomimetics may be expected to increase gastric acid secretion due to increased cholinergic activity. Therefore, patients should be monitored closely for symptoms of active or occult gastrointestinal bleeding, especially those with an increased risk for developing ulcers, e.g., those with a history of ulcer disease or patients using concurrent nonsteroidal anti-inflammatory drugs (NSAIDs). Clinical studies of galantamine have shown no increase, relative to placebo, in the incidence of either peptic ulcer disease or gastrointestinal bleeding.
Galantamine, as a predictable consequence of its pharmacological properties, has been shown to produce nausea, vomiting, diarrhea, anorexia, and weight loss. During therapy, the patient’s weight should be monitored [see Adverse Reactions (6.1)].
5.4 Genitourinary Conditions
Although this was not observed in clinical trials with galantamine, cholinomimetics may cause bladder outflow obstruction.
5.5 Neurological Conditions
Seizures: Cholinesterase inhibitors are believed to have some potential to cause generalized convulsions. However, seizure activity may also be a manifestation of Alzheimer’s disease. In clinical trials, there was no increase in the incidence of convulsions with galantamine compared to placebo. Patients with Alzheimer’s disease should be monitored closely for seizures while taking galantamine.
5.6 Pulmonary Conditions
Because of its cholinomimetic action, galantamine should be prescribed with care to patients with a history of severe asthma or obstructive pulmonary disease. Respiratory function should be monitored closely for the occurrence of respiratory adverse effects.
5.7 Deaths in Subjects With Mild Cognitive Impairment (MCI)
In two randomized placebo controlled trials of 2 years duration in subjects with mild cognitive impairment (MCI), a total of 13 subjects on galantamine (n = 1026) and 1 subject on placebo (n = 1022) died. The deaths were due to various causes which could be expected in an elderly population; about half of the galantamine deaths appeared to result from various vascular causes (myocardial infarction, stroke, and sudden death).
Although the difference in mortality between galantamine- and placebo-treated groups in these two studies was significant, the results are highly discrepant with other studies of galantamine. Specifically, in these two MCI studies, the mortality rate in the placebo-treated subjects was markedly lower than the rate in placebo-treated patients in trials of galantamine in Alzheimer’s disease or other dementias (0.7 per 1000 person years compared to 22 to 61 per 1000 person years, respectively). Although the mortality rate in the galantamine-treated MCI subjects was also lower than that observed in galantamine-treated patients in Alzheimer’s disease and other dementia trials (10.2 per 1000 person years compared to 23 to 31 per 1000 person years, respectively), the relative difference was much less. When the Alzheimer’s disease and other dementia studies were pooled (n = 6000), the mortality rate in the placebo group numerically exceeded that in the galantamine group. Furthermore, in the MCI studies, no subjects in the placebo group died after 6 months, a highly unexpected finding in this population.
Individuals with mild cognitive impairment demonstrate isolated memory impairment greater than expected for their age and education, but do not meet current diagnostic criteria for Alzheimer’s disease.
6 ADVERSE REACTIONS
The following are discussed in more detail in other sections of the labeling:
- Anesthesia [see Warnings and Precautions (5.1)]
- Cardiovascular conditions [see Warnings and Precautions (5.2)]
- Gastrointestinal conditions [see Warnings and Precautions (5.3)]
- Genitourinary conditions [see Warnings and Precautions (5.4)]
- Neurological conditions [see Warnings and Precautions (5.5)]
- Pulmonary conditions [see Warnings and Precautions (5.6)]
- Deaths in subjects with mild cognitive impairment (MCI) [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions in galantamine-treated subjects from double-blind clinical trials (≥ 5%) were nausea, vomiting, diarrhea, dizziness, headache, decreased appetite, and weight decreased.
The most common adverse reactions associated with discontinuation (≥ 1%) in galantamine-treated subjects from double-blind clinical trials were nausea (7.7%), vomiting (4.1%), decreased appetite (1.9%), and dizziness (1.6%).
The safety of the extended-release capsule and immediate-release tablet formulations of galantamine was evaluated in 2932 galantamine-treated subjects who participated in 7 placebo-controlled clinical studies and 1454 subjects in 5 open-label clinical studies with mild to moderate dementia of the Alzheimer’s type. In clinical studies, the safety profile of once-daily treatment with extended-release galantamine was similar in frequency and nature to that seen with tablets. The information presented in this section was derived from pooled double-blind studies and from pooled open-label data.
Throughout this section, adverse reactions are reported. Adverse reactions are adverse events that were considered to be reasonably associated with the use of galantamine based on the comprehensive assessment of the available adverse event information. A causal relationship with galantamine cannot be reliably established in individual cases.
Commonly-Observed Adverse Reactions in Double-Blind, Placebo-Controlled Clinical Trials
| Galantamine | Placebo | |
| System/Organ Class | (n = 2932) | (n = 1525) |
| Adverse Reaction | % | % |
| Metabolism And Nutrition Disorders | ||
| Decreased Appetite | 5.2 | 1.4 |
| Anorexia | 3.8 | 1 |
| Psychiatric Disorders | ||
| Depression | 4.2 | 2.9 |
| Nervous System Disorders | ||
| Dizziness | 8.9 | 4.6 |
| Headache | 7.6 | 5.4 |
| Tremor | 2 | 0.8 |
| Syncope | 1.8 | 0.7 |
| Lethargy | 1.7 | 0.7 |
| Somnolence | 1.7 | 0.8 |
| Cardiac Disorders | ||
| Bradycardia | 1.2 | 0.3 |
| Gastrointestinal Disorders | ||
| Nausea | 25 | 7.6 |
| Vomiting | 12.8 | 3.1 |
| Diarrhea | 9 | 6.3 |
| Abdominal Pain | 2.4 | 0.9 |
| Abdominal Pain Upper | 2 | 1.4 |
| Dyspepsia | 1.8 | 1.3 |
| Stomach Discomfort | 1.6 | 0.6 |
| Abdominal Discomfort | 1 | 0.4 |
| Skin And Subcutaneous Tissue Disorders | ||
| Hyperhidrosis | 1.2 | 0.7 |
| Musculoskeletal And Connective Tissue Disorders | ||
| Muscle Spasms | 1.5 | 0.8 |
| General Disorders And Administration Site Conditions | ||
| Fatigue | 4 | 2.2 |
| Asthenia | 2.3 | 1.7 |
| Malaise | 1.4 | 0.7 |
| Investigations | ||
| Weight Decreased | 5.1 | 1.4 |
The majority of these adverse events occurred during the dose-escalation period. In those patients who experienced the most frequent adverse event, nausea, the median duration of the nausea was 5 to 7 days.
Administration of galantamine hydrobromide with food, the use of anti-emetic medication, and ensuring adequate fluid intake may reduce the impact of these events [see Dosage and Administration (2) and Patient Counseling Information (17)].
Other Adverse Reactions Observed in Clinical Trials of Galantamine
The following adverse reactions occurred in < 1% of all galantamine-treated subjects (N = 2932) in the above double-blind, placebo-controlled clinical trial data sets. In addition, the following also includes all adverse reactions reported at any frequency rate in subjects (N = 1454) who participated in open-label studies. Adverse reactions listed in Table 1 above were not included below:
Metabolism and Nutrition Disorders: Dehydration
Nervous System Disorders: Dysgeusia, Hypersomnia, Paresthesia
Eye Disorders: Vision blurred
Cardiac Disorders: Atrioventricular block first degree, Palpitations, Sinus bradycardia, Supraventricular extrasystoles
Vascular Disorders: Flushing, Hypotension
Gastrointestinal Disorders: Retching
Musculoskeletal and Connective Tissue Disorders: Muscular weakness
Injury, Poisoning and Procedural Complications: Fall
Discontinuations Due to Adverse Reactions
In the 7 placebo-controlled studies of adults, 379 (12.9%) galantamine-treated patients (N = 2932) and 42 (2.8%) placebo patients (N = 1525) discontinued due to an adverse reaction. Those events with an incidence of ≥ 0.5% in the galantamine-treated patients included nausea (225, 7.7%), vomiting (119, 4.1%), decreased appetite (56, 1.9%), dizziness (48, 1.6%), diarrhea (27, 0.9%), headache (26, 0.9%) weight decreased (24, 0.8%), and abdominal pain (15, 0.5%). Those events with an incidence of ≥ 0.5% in placebo patients included nausea (14, 0.9%) and dizziness (8, 0.5%).
In the 5 open-label studies, 103 (7.1%) patients (N = 1454) discontinued due to an adverse reaction. Those events with an incidence of ≥ 0.5% included nausea (43, 3%), vomiting (23, 1.6%), decreased appetite (13, 0.9%), headache (12, 0.8%), weight decreased (9, 0.6%), dizziness (8, 0.6%), and diarrhea (7, 0.5%).
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of galantamine hydrobromide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency:
Immune System Disorders: Hypersensitivity
Psychiatric Disorders: Hallucination, Hallucination visual, Hallucination auditory
Ear and Labyrinth Disorders: Tinnitus
Vascular Disorders: Hypertension
Hepatobiliary Disorders: Hepatitis, Hepatic enzyme increased
7 DRUG INTERACTIONS
7.1 Use With Anticholinergics
Galantamine has the potential to interfere with the activity of anticholinergic medications [see Clinical Pharmacology (12.3)].
7.2 Use With Cholinomimetics and Other Cholinesterase Inhibitors
A synergistic effect is expected when cholinesterase inhibitors are given concurrently with succinylcholine, other cholinesterase inhibitors, similar neuromuscular blocking agents or cholinergic agonists such as bethanechol [see Clinical Pharmacology (12.3)].
7.3 Effect of Other Drugs on Galantamine
In Vitro
CYP3A4 and CYP2D6 are the major enzymes involved in the metabolism of galantamine. CYP3A4 mediates the formation of galantamine-N-oxide; CYP2D6 leads to the formation of O-desmethyl-galantamine. Because galantamine is also glucuronidated and excreted unchanged, no single pathway appears predominant [see Clinical Pharmacology (12.3)].
In Vivo
Cimetidine and Ranitidine
Galantamine was administered as a single dose of 4 mg on day 2 of a 3 day treatment with either cimetidine (800 mg daily) or ranitidine (300 mg daily). Cimetidine increased the bioavailability of galantamine by approximately 16%. Ranitidine had no effect on the pharmacokinetics of galantamine.
Ketoconazole
Ketoconazole, a strong inhibitor of CYP3A4 and an inhibitor of CYP2D6, at a dose of 200 mg BID for 4 days, increased the AUC of galantamine by 30%.
Erythromycin
Erythromycin, a moderate inhibitor of CYP3A4 at a dose of 500 mg QID for 4 days, affected the AUC of galantamine minimally (10% increase).
Paroxetine
Paroxetine, a strong inhibitor of CYP2D6, at 20 mg/day for 16 days, increased the oral bioavailability of galantamine by about 40%.
Memantine
Memantine, an N-methyl-D-aspartate receptor antagonist, at a dose of 10 mg BID, had no effect on the pharmacokinetics of galantamine (16 mg/day) at steady state.
7.4 Effect of Galantamine on Other Drugs
In Vitro
Galantamine did not inhibit the metabolic pathways catalyzed by CYP1A2, CYP2A6, CYP3A4, CYP4A, CYP2C, CYP2D6 or CYP2E1. This indicates that the inhibitory potential of galantamine towards the major forms of cytochrome P450 is very low [see Clinical Pharmacology (12.3)].
In Vivo
Warfarin
Galantamine at 24 mg/day had no effect on the pharmacokinetics of R- and S-warfarin (25 mg single dose) or on the prothrombin time. The protein binding of warfarin was unaffected by galantamine.
Digoxin
Galantamine at 24 mg/day had no effect on the steady-state pharmacokinetics of digoxin (0.375 mg once daily) when they were coadministered. In this study, however, one healthy subject was hospitalized for 2nd and 3rd degree heart block and bradycardia.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects
Pregnancy Category C
There are no adequate and well-controlled studies in pregnant women. In studies conducted in animals, administration of galantamine during pregnancy resulted in developmental toxicity (increased incidence of morphological abnormalities and decreased growth in offspring) at doses similar to or greater than those used clinically. Galantamine hydrobromide should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In rats, administration of galantamine (oral doses of 2, 8, or 16 mg/kg/day), from day 14 (females) or day 60 (males) prior to mating and continuing in females through the period of organogenesis, resulted in an increased incidence of fetal skeletal variations at the two highest doses. The no-effect dose for embryo-fetal developmental toxicity in rats (2 mg/kg/day) is approximately equal to the maximum recommended human dose (MRHD of 24 mg/day) on a body surface area (mg/m2) basis. When galantamine (oral doses of 4, 12, 28, or 48 mg/kg/day) was administered to pregnant rabbits throughout the period of organogenesis, small increases in fetal visceral malformations and skeletal variations were observed at the highest dose. The no-effect dose for embryo-fetal developmental toxicity in rabbits (28 mg/kg/day) is approximately 20 times the MRHD on a mg/m2 basis. In a study in which pregnant rats were orally dosed with galantamine (2, 8, or 16 mg/kg/day) from the beginning of organogenesis through day 21 post-partum, pup weights were decreased at birth and during the lactation period at the two highest doses. The no-effect dose for pre- and postnatal developmental toxicity in rats (2 mg/kg/day) is approximately equal to the MRHD on a mg/m2 basis.
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when galantamine hydrobromide is administered to a nursing woman.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Alzheimer’s disease is a disorder occurring primarily in individuals over 55 years of age. Seven double-blind, placebo-controlled clinical trials and 5 open-label trials in a total of 4474 patients have investigated galantamine hydrobromide in the treatment of mild to moderate dementia of the Alzheimer’s type [see Adverse Reactions (6.1) and Clinical Studies (14)]. The mean age of patients enrolled in these clinical studies was 75 years; 78% of these patients were between 65 and 84 years of age, and 11% of patients were 85 years of age or older.
8.6 Hepatic Impairment
In patients with moderately impaired hepatic function, dose titration should proceed cautiously [see Clinical Pharmacology (12.3) and Dosage and Administration (2.3)]. The use of galantamine hydrobromide in patients with severe hepatic impairment is not recommended.
8.7 Renal Impairment
In patients with moderately impaired renal function, dose titration should proceed cautiously [see Clinical Pharmacology (12.3) and Dosage and Administration (2.4)]. In patients with severely impaired renal function (CLcr < 9 mL/min) the use of galantamine hydrobromide is not recommended.
10 OVERDOSAGE
Because strategies for the management of overdose are continually evolving, it is advisable to contact a poison control center to determine the latest recommendations for the management of an overdose of any drug.
As in any case of overdose, general supportive measures should be utilized. Signs and symptoms of significant overdosing of galantamine are predicted to be similar to those of overdosing of other cholinomimetics. These effects generally involve the central nervous system, the parasympathetic nervous system, and the neuromuscular junction. In addition to muscle weakness or fasciculations, some or all of the following signs of cholinergic crisis may develop: severe nausea, vomiting, gastrointestinal cramping, salivation, lacrimation, urination, defecation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved.
Tertiary anticholinergics such as atropine may be used as an antidote for galantamine hydrobromide overdosage. Intravenous atropine sulfate titrated to effect is recommended at an initial dose of 0.5 to 1 mg i.v. with subsequent doses based upon clinical response. Atypical responses in blood pressure and heart rate have been reported with other cholinomimetics when coadministered with quaternary anticholinergics. It is not known whether galantamine and/or its metabolites can be removed by dialysis (hemodialysis, peritoneal dialysis, or hemofiltration). Dose-related signs of toxicity in animals included hypoactivity, tremors, clonic convulsions, salivation, lacrimation, chromodacryorrhea, mucoid feces, and dyspnea.
In one postmarketing report, one patient who had been taking 4 mg of galantamine daily for a week inadvertently ingested eight 4 mg tablets (32 mg total) on a single day. Subsequently, she developed bradycardia, QT prolongation, ventricular tachycardia and torsades de pointes accompanied by a brief loss of consciousness for which she required hospital treatment. Two additional cases of accidental ingestion of 32 mg (nausea, vomiting, and dry mouth; nausea, vomiting, and substernal chest pain) and one of 40 mg (vomiting), resulted in brief hospitalizations for observation with full recovery. One patient, who was prescribed 24 mg/day and had a history of hallucinations over the previous two years, mistakenly received 24 mg twice daily for 34 days and developed hallucinations requiring hospitalization. Another patient, who was prescribed 16 mg/day of oral solution, inadvertently ingested 160 mg (40 mL) and experienced sweating, vomiting, bradycardia, and near-syncope one hour later, which necessitated hospital treatment. His symptoms resolved within 24 hours.
11 DESCRIPTION
Galantamine hydrobromide, USP is a reversible, competitive acetylcholinesterase inhibitor. Galantamine hydrobromide, USP is known chemically as (4aS,6R,8aS)-4a,5,9,10,11,12-hexahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol hydrobromide. Galantamine hydrobromide is a white to almost white powder and is sparingly soluble in water. The structural formula for galantamine hydrobromide is:

C17H21NO3 ·HBr Molecular Weight: 368.27
Galantamine Hydrobromide Extended-Release Capsules are available in opaque hard gelatin capsules of 8 mg (light beige), 16 mg (pink), and 24 mg (tan) containing galantamine hydrobromide, USP equivalent to 8, 16, and 24 mg galantamine base respectively.
Inactive ingredients are: D&C yellow no. 10 aluminum lake, dibutyl sebacate, ethylcellulose, FD&C blue no. 1 aluminum lake, FD&C blue no. 2 aluminum lake, FD&C red no. 40 aluminum lake, gelatin, hypromellose, pharmaceutical glaze, propylene glycol, sugar spheres, synthetic black iron oxide, talc and titanium dioxide.
In addition: the 8 mg capsule also contains black iron oxide, red iron oxide and yellow iron oxide. The 16 mg capsule also contains FD&C red no. 40. The 24 mg capsule also contains D&C yellow no. 10, FD&C blue no. 1 and FD&C red no. 40.
Galantamine Tablets USP for oral use are available in round, biconvex, film-coated tablets of 4 mg (yellow), 8 mg (dark pink), and 12 mg (red). Each 4, 8, and 12 mg (base equivalent) tablet contains 5.126, 10.253, and 15.379 mg of galantamine hydrobromide, respectively.
Inactive ingredients are: hypromellose, mannitol, polyethylene glycol, polysorbate 80, sodium stearyl fumarate, sodium starch glycolate, talc and titanium dioxide.
In addition: the 4 mg also contains D&C yellow no. 10 aluminum lake and FD&C yellow no. 6/Sunset yellow FCF aluminum lake. The 8 mg also contains D&C red no. 30/Helendon pink aluminum lake and FD&C blue no. 2/Indigo carmine aluminum lake. The 12 mg strength also contains FD&C red no. 40/Allura red AC aluminum lake.
Tablets meet USP Dissolution Test 2.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Although the etiology of cognitive impairment in Alzheimer’s disease (AD) is not fully understood, it has been reported that acetylcholine-producing neurons degenerate in the brains of patients with Alzheimer’s disease. The degree of this cholinergic loss has been correlated with degree of cognitive impairment and density of amyloid plaques (a neuropathological hallmark of Alzheimer’s disease).
Galantamine, a tertiary alkaloid, is a competitive and reversible inhibitor of acetylcholinesterase. While the precise mechanism of galantamine’s action is unknown, it is postulated to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase. If this mechanism is correct, galantamine’s effect may lessen as the disease process advances and fewer cholinergic neurons remain functionally intact. There is no evidence that galantamine alters the course of the underlying dementing process.
12.3 Pharmacokinetics
Galantamine is well absorbed with absolute oral bioavailability of about 90%. It has a terminal elimination half-life of about 7 hours and pharmacokinetics are linear over the range of 8 to 32 mg/day.
The maximum inhibition of acetylcholinesterase activity of about 40% was achieved about one hour after a single oral dose of 8 mg galantamine in healthy male subjects.
Absorption and Distribution
Galantamine is rapidly and completely absorbed with time to peak concentration about 1 hour. Bioavailability of the tablet was the same as the bioavailability of an oral solution. Food did not affect the AUC of galantamine but Cmax decreased by 25% and Tmax was delayed by 1.5 hours. The mean volume of distribution of galantamine is 175 L.
The plasma protein binding of galantamine is 18% at therapeutically relevant concentrations. In whole blood, galantamine is mainly distributed to blood cells (52.7%). The blood to plasma concentration ratio of galantamine is 1.2.
Metabolism and Elimination
Galantamine is metabolized by hepatic cytochrome P450 enzymes, glucuronidated, and excreted unchanged in the urine. In vitro studies indicate that cytochrome CYP2D6 and CYP3A4 were the major cytochrome P450 isoenzymes involved in the metabolism of galantamine, and inhibitors of both pathways increase oral bioavailability of galantamine modestly [see Drug Interactions (7.3)]. O-demethylation, mediated by CYP2D6 was greater in extensive metabolizers of CYP2D6 than in poor metabolizers. In plasma from both poor and extensive metabolizers, however, unchanged galantamine and its glucuronide accounted for most of the sample radioactivity.
In studies of oral 3H-galantamine, unchanged galantamine and its glucuronide, accounted for most plasma radioactivity in poor and extensive CYP2D6 metabolizers. Up to 8 hours post-dose, unchanged galantamine accounted for 39 to 77% of the total radioactivity in the plasma, and galantamine glucuronide for 14 to 24%. By 7 days, 93 to 99% of the radioactivity had been recovered, with about 95% in urine and about 5% in the feces. Total urinary recovery of unchanged galantamine accounted for, on average, 32% of the dose and that of galantamine glucuronide for another 12% on average.
After i.v. or oral administration, about 20% of the dose was excreted as unchanged galantamine in the urine in 24 hours, representing a renal clearance of about 65 mL/min, about 20 to 25% of the total plasma clearance of about 300 mL/min.
Galantamine hydrobromide 24 mg extended-release capsules administered once daily under fasting conditions are bioequivalent to galantamine tablets 12 mg twice daily with respect to AUC24h and Cmin. The Cmax and Tmax of the extended-release capsules were lower and occurred later, respectively, compared with the immediate-release tablets, with Cmax about 25% lower and median Tmax occurring about 4.5 to 5 hours after dosing. Dose-proportionality is observed for galantamine hydrobromide extended-release capsules over the dose range of 8 to 24 mg daily and steady state is achieved within a week. There was no effect of age on the pharmacokinetics of galantamine hydrobromide extended-release capsules. CYP2D6 poor metabolizers had drug exposures that were approximately 50% higher than for extensive metabolizers.
There are no appreciable differences in pharmacokinetic parameters when galantamine hydrobromide extended-release capsules are given with food compared to when they are given in the fasted state.
Specific Populations
CYP2D6 Poor Metabolizers
Approximately 7% of the normal population has a genetic variation that leads to reduced levels of activity of CYP2D6 isozyme. Such individuals have been referred to as poor metabolizers. After a single oral dose of 4 mg or 8 mg galantamine, CYP2D6 poor metabolizers demonstrated a similar Cmax and about 35% AUC∞ increase of unchanged galantamine compared to extensive metabolizers.
A total of 356 patients with Alzheimer’s disease enrolled in two Phase 3 studies were genotyped with respect to CYP2D6 (n = 210 hetero-extensive metabolizers, 126 homo-extensive metabolizers, and 20 poor metabolizers). Population pharmacokinetic analysis indicated that there was a 25% decrease in median clearance in poor metabolizers compared to extensive metabolizers. Dosage adjustment is not necessary in patients identified as poor metabolizers as the dose of drug is individually titrated to tolerability.
Hepatic Impairment
Following a single 4 mg dose of galantamine tablets, the pharmacokinetics of galantamine in subjects with mild hepatic impairment (n = 8; Child-Pugh score of 5 to 6) were similar to those in healthy subjects. In patients with moderate hepatic impairment (n = 8; Child-Pugh score of 7 to 9), galantamine clearance was decreased by about 25% compared to normal volunteers. Exposure would be expected to increase further with increasing degree of hepatic impairment [see Use in Specific Populations (8.6) and Dosage and Administration (2.3)].
Renal Impairment
Following a single 8 mg dose of galantamine tablets, AUC increased by 37% and 67% in moderate and severely renal-impaired patients compared to normal volunteers [see Use in Specific Populations (8.7) and Dosage and Administration (2.4)].
Elderly
Data from clinical trials in patients with Alzheimer’s disease indicate that galantamine concentrations are 30 to 40% higher than in healthy young subjects.
Gender and Race
No specific pharmacokinetic study was conducted to investigate the effect of gender and race on the disposition of galantamine, but a population pharmacokinetic analysis indicates (n = 539 males and 550 females) that galantamine clearance is about 20% lower in females than in males (explained by lower body weight in females) and race (n = 1029 White, 24 Black, 13 Asian and 23 other) did not affect the clearance of galantamine.
Drug-Drug Interactions
Multiple metabolic pathways and renal excretion are involved in the elimination of galantamine so no single pathway appears predominant. Based on in vitro studies, CYP2D6 and CYP3A4 were the major enzymes involved in the metabolism of galantamine. CYP2D6 was involved in the formation of O-desmethyl-galantamine, whereas CYP3A4 mediated the formation of galantamine-N-oxide. Galantamine is also glucuronidated and excreted unchanged in urine.
Effect of Other Drugs on the Metabolism of Galantamine
Drugs that are potent inhibitors for CYP2D6 or CYP3A4 may increase the AUC of galantamine. Multiple dose pharmacokinetic studies demonstrated that the AUC of galantamine increased 30% and 40%, respectively, during coadministration of ketoconazole and paroxetine. As coadministered with erythromycin, another CYP3A4 inhibitor, the galantamine AUC increased only 10%. Population pharmacokinetics analysis with a database of 852 patients with Alzheimer’s disease showed that the clearance of galantamine was decreased about 25 to 33% by concurrent administration of amitriptyline (n = 17), fluoxetine (n = 48), fluvoxamine (n = 14), and quinidine (n = 7), known inhibitors of CYP2D6.
Concurrent administration of H2-antagonists demonstrated that ranitidine did not affect the pharmacokinetics of galantamine, and cimetidine increased the galantamine AUC by approximately 16%.
A multiple dose pharmacokinetic study with concurrent administration of memantine, an N-methyl-D-aspartate (NMDA) receptor antagonist, demonstrated that coadministration of memantine in a dose of 10 mg BID did not affect the pharmacokinetic profile of galantamine (16 mg daily) at steady state.
Effect of Galantamine on the Metabolism of Other Drugs
In vitro studies show that galantamine did not inhibit the metabolic pathways catalyzed by CYP1A2, CYP2A6, CYP3A4, CYP4A, CYP2C, CYP2D6 or CYP2E1. This indicates that the inhibitory potential of galantamine towards the major forms of cytochrome P450 is very low. Multiple doses of galantamine (24 mg/day) had no effect on the pharmacokinetics of digoxin and warfarin (R- and S- forms). Galantamine had no effect on the increased prothrombin time induced by warfarin.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a 24 month oral carcinogenicity study in rats, an increase in endometrial adenocarcinomas was observed at 10 mg/kg/day (4 times the MRHD of 24 mg/day on a mg/m2 basis or 6 times on a plasma exposure [AUC] basis) and 30 mg/kg/day (12 times MRHD on a mg/m2 basis or 19 times on an AUC basis). No increase in neoplastic changes was observed in females at 2.5 mg/kg/day (equivalent to the MRHD on a mg/m2 basis or 2 times on an AUC basis) or in males up to the highest dose tested of 30 mg/kg/day (12 times the MRHD on a mg/m2 and AUC basis).
Galantamine was not carcinogenic in a 6 month carcinogenicity study in transgenic (P 53-deficient) mice at oral doses up to 20 mg/kg/day, or in a 24 month carcinogenicity study in mice at oral doses up to 10 mg/kg/day (equivalent to the MRHD on a plasma AUC basis).
Mutagenesis
Galantamine was negative in a battery of in vitro (bacterial reverse mutation, mouse lymphoma tk, and chromosomal aberration in mammalian cells) and in vivo (mouse micronucleus) genotoxicity assays.
Impairment of Fertility
No impairment of fertility was seen in rats given up to 16 mg/kg/day (7 times the MRHD on a mg/m2 basis) for 14 days prior to mating in females and for 60 days prior to mating in males.
14 CLINICAL STUDIES
The effectiveness of galantamine as a treatment for Alzheimer’s disease is demonstrated by the results of 5 randomized, double-blind, placebo-controlled clinical investigations in patients with probable Alzheimer’s disease, 4 with the immediate-release tablet and 1 with the extended-release capsule [diagnosed by NINCDS-ADRDA criteria, with Mini-Mental State Examination scores that were ≥ 10 and ≤ 24]. Doses studied with the tablet formulation were 8 to 32 mg/day given as twice daily doses. In 3 of the 4 studies with the tablet, patients were started on a low dose of 8 mg, then titrated weekly by 8 mg/day to 24 or 32 mg as assigned. In the fourth study (USA 4 week Dose Escalation Fixed-Dose Study) dose escalation of 8 mg/day occurred over 4 week intervals. The mean age of patients participating in these 4 galantamine trials was 75 years with a range of 41 to 100. Approximately 62% of patients were women and 38% were men. The racial distribution was White 94%, Black 3% and other races 3%. Two other studies examined a three times daily dosing regimen; these also showed or suggested benefit but did not suggest an advantage over twice daily dosing.
14.1 Study Outcome Measures
In each study, the primary effectiveness of galantamine was evaluated using a dual outcome assessment strategy as measured by the Alzheimer’s Disease Assessment Scale (ADAS-cog) and the Clinician’s Interview Based Impression of Change that required the use of caregiver information (CIBIC-plus).
The ability of galantamine to improve cognitive performance was assessed with the cognitive sub-scale of the Alzheimer’s Disease Assessment Scale (ADAS-cog), a multi-item instrument that has been extensively validated in longitudinal cohorts of Alzheimer’s disease patients. The ADAS-cog examines selected aspects of cognitive performance including elements of memory, orientation, attention, reasoning, language and praxis. The ADAS-cog scoring range is from 0 to 70, with higher scores indicating greater cognitive impairment. Elderly normal adults may score as low as 0 or 1, but it is not unusual for non-demented adults to score slightly higher.
The patients recruited as participants in each study using the tablet formulation had mean scores on ADAS-cog of approximately 27 units, with a range from 5 to 69. Experience gained in longitudinal studies of ambulatory patients with mild to moderate Alzheimer’s disease suggests that they gain 6 to 12 units a year on the ADAS-cog. Lesser degrees of change, however, are seen in patients with very mild or very advanced disease because the ADAS-cog is not uniformly sensitive to change over the course of the disease. The annualized rate of decline in the placebo patients participating in galantamine trials was approximately 4.5 units per year.
The ability of galantamine to produce an overall clinical effect was assessed using a Clinician’s Interview Based Impression of Change that required the use of caregiver information, the CIBIC-plus. The CIBIC-plus is not a single instrument and is not a standardized instrument like the ADAS-cog. Clinical trials for investigational drugs have used a variety of CIBIC formats, each different in terms of depth and structure. As such, results from a CIBIC-plus reflect clinical experience from the trial or trials in which it was used and cannot be compared directly with the results of CIBIC-plus evaluations from other clinical trials. The CIBIC-plus used in the trials was a semi-structured instrument based on a comprehensive evaluation at baseline and subsequent time-points of 4 major areas of patient function: general, cognitive, behavioral and activities of daily living. It represents the assessment of a skilled clinician based on his/her observation at an interview with the patient, in combination with information supplied by a caregiver familiar with the behavior of the patient over the interval rated. The CIBIC-plus is scored as a seven point categorical rating, ranging from a score of 1, indicating “markedly improved,” to a score of 4, indicating “no change” to a score of 7, indicating “marked worsening.” The CIBIC-plus has not been systematically compared directly to assessments not using information from caregivers (CIBIC) or other global methods.
14.2 Immediate-Release Tablets
U.S. Twenty-One Week Fixed-Dose Study
In a study of 21 weeks duration, 978 patients were randomized to doses of 8, 16, or 24 mg of galantamine per day, or to placebo, each given in 2 divided doses. Treatment was initiated at 8 mg/day for all patients randomized to galantamine, and increased by 8 mg/day every 4 weeks. Therefore, the maximum titration phase was 8 weeks and the minimum maintenance phase was 13 weeks (in patients randomized to 24 mg/day of galantamine).
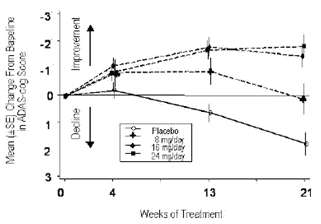
Effects on the ADAS-cog
Figure 1 illustrates the time course for the change from baseline in ADAS-cog scores for all four dose groups over the 21 weeks of the study. At 21 weeks of treatment, the mean differences in the ADAS-cog change scores for the galantamine-treated patients compared to the patients on placebo were 1.7, 3.3, and 3.6 units for the 8, 16 and 24 mg/day treatments, respectively. The 16 mg/day and 24 mg/day treatments were statistically significantly superior to placebo and to the 8 mg/day treatment. There was no statistically significant difference between the 16 mg/day and 24 mg/day dose groups.
Age, Gender and Race
Patient’s age, gender, or race did not predict clinical outcome of treatment.
14.3 Extended-Release Capsules
The efficacy of galantamine extended-release capsules was studied in a randomized, double-blind, placebo-controlled trial which was 6 months in duration, and had an initial 4 week dose-escalation phase. In this trial, patients were assigned to one of 3 treatment groups: Galantamine extended-release capsules in a flexible dose of 16 to 24 mg once daily; galantamine tablets in a flexible dose of 8 to 12 mg twice daily; and placebo. The primary efficacy measures in this study were the ADAS-cog and CIBIC-plus. On the protocol-specified primary efficacy analysis at Month 6, a statistically significant improvement favoring galantamine extended-release capsules over placebo was seen for the ADAS-cog, but not for the CIBIC-plus. Galantamine extended-release capsules showed a statistically significant improvement when compared with placebo on the Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL) scale, a measure of function, and a secondary efficacy measure in this study. The effects of both galantamine extended-release capsules and galantamine tablets on the ADAS-cog, CIBIC-plus, and ADCS-ADL were similar in this study.
16 HOW SUPPLIED/STORAGE AND HANDLING
Galantamine Hydrobromide Extended-Release Capsules are supplied as follows:
8 mg: Light beige opaque hard gelatin capsules filled with white to off-white pellets, imprinted barr over 8 mg and 1020 in black ink. They are packaged in unit-of-use bottles of 30 capsules.
16 mg: Pink opaque hard gelatin capsules filled with white to off-white pellets, imprinted barr over 16 mg and 1021 in black ink. They are packaged in unit-of-use bottles of 30 capsules.
24 mg: Tan opaque hard gelatin capsules filled with white to off-white pellets, imprinted barr over 24 mg and 1022 in black ink. They are packaged in unit-of-use bottles of 30 capsules.
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Galantamine Tablets USP are supplied as follows:
4 mg: Yellow, round, film-coated, biconvex, unscored tablets, debossed with “stylized b” 4 on one side and 138 on the other side. They are packaged in bottles of 60 tablets.
8 mg: Dark pink, round, film-coated, biconvex, unscored tablets, debossed with “stylized b” 8 on one side and 139 on the other side. They are packaged in bottles of 60 tablets.
12 mg: Red, round, film-coated, biconvex, unscored tablets, debossed with “stylized b” 12 on one side and 140 on the other side. They are packaged in bottles of 60 tablets.
Storage and Handling
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
17 PATIENT COUNSELING INFORMATION
General Dosing Guidance
Caregivers should be instructed about the recommended dosage and administration of galantamine hydrobromide extended-release capsules and galantamine tablets. Galantamine hydrobromide extended-release capsules should be administered once daily in the morning, preferably with food. Galantamine tablets should be administered twice per day, preferably with the morning and evening meals. Dose escalation (dose increases) should follow a minimum of four weeks at prior dose. If therapy has been interrupted for more than three days, the patient should be restarted with the lowest dose and then re-titrated to an appropriate dosage [see Dosage and Administration (2)].
Patients and caregivers should be advised to ensure adequate fluid intake during treatment [see Dosage and Administration (2)].
Patients and caregivers should be advised that the most frequent adverse events associated with use of the drug can be minimized by following the recommended dosage and administration.
TEVA PHARMACEUTICALS USA
Sellersville, PA 18960
Rev. D 8/2013
PRINCIPAL DISPLAY PANEL

Galantamine Hydrobromide Extended-Release Capsules 8 mg 30s Label Text
NDC 0555-1020-01
GALANTAMINE
HYDROBROMIDE
Extended-Release
Capsules
8 mg*
Rx only
30 CAPSULES (Unit-of-Use)
TEVA
PRINCIPAL DISPLAY PANEL

Galantamine Hydrobromide Extended-Release Capsules 16 mg 30s Label Text
NDC 0555-1021-01
GALANTAMINE
HYDROBROMIDE
Extended-Release
Capsules
16 mg*
Rx only
30 CAPSULES (Unit-of-Use)
TEVA
PRINCIPAL DISPLAY PANEL

Galantamine Hydrobromide Extended-Release Capsules 24 mg 30s Label Text
NDC 0555-1022-01
GALANTAMINE
HYDROBROMIDE
Extended-Release
Capsules
24 mg*
Rx only
30 CAPSULES (Unit-of-Use)
TEVA
PRINCIPAL DISPLAY PANEL

Galantamine Tablets USP 4 mg 60s Label Text
NDC 0555-0138-09
GALANTAMINE
Tablets USP
4 mg*
Rx only
60 TABLETS
TEVA
PRINCIPAL DISPLAY PANEL

Galantamine Tablets USP 8 mg 60s Label Text
NDC 0555-0139-09
GALANTAMINE
Tablets USP
8 mg*
Rx only
60 TABLETS
TEVA
PRINCIPAL DISPLAY PANEL

Galantamine Tablets USP 12 mg 60s Label Text
NDC 0555-0140-09
GALANTAMINE
Tablets USP
12 mg*
Rx only
60 TABLETS
TEVA
Galantamine HydrobromideGalantamine Hydrobromide CAPSULE, EXTENDED RELEASE
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Galantamine HydrobromideGalantamine Hydrobromide CAPSULE, EXTENDED RELEASE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Galantamine HydrobromideGalantamine Hydrobromide CAPSULE, EXTENDED RELEASE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
GalantamineGalantamine TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
GalantamineGalantamine TABLET, FILM COATED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
GalantamineGalantamine TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||