Eye Drops AC
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredients
- Purpose
- Keep out of reach of children.
- Eye Drops AC Uses
- Warnings
- Directions
- Inactive Ingredients
- Eye Drops AC Other information
- If pregnant or breast-feeding,
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Active Ingredients
Tetrahydrozoline HCl 0.05%
Zinc sulfate 0.25%
Purpose
Tetrahydrozoline HCl - Redness reliever
Zinc sulfate - Astringent
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Eye Drops AC Uses
- for temporary relief of discomfort and redness of the eye due to minor eye irritations
Warnings
For external use only
Ask a doctor before use if you have narrow angle glaucoma
When using this product
- pupils may become enlarged temporarily
- to avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- if solution changes color or becomes cloudy, do not use
- overuse may produce increased redness of the eye
- remove contact lens before using
Stop use and ask a doctor if you experience
- eye pain
- changes in vision
- continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours
Directions
Instill 1 or 2 drops in the affected eye(s) up to four times daily.
Inactive Ingredients
benzalkonium chloride, boric acid, edetate disodium, purified water, sodium chloride, and sodium citrate
Eye Drops AC Other information
- Tamper Evident. Do not use this product if imprinted neckband is missing or broken.
- RETAIN THIS CARTON FOR FUTURE REFERENCE
- Store at 15o - 30oC (59o - 86oF)
If pregnant or breast-feeding,
ask a health professional before use.
Principal Display Panel
EDAC
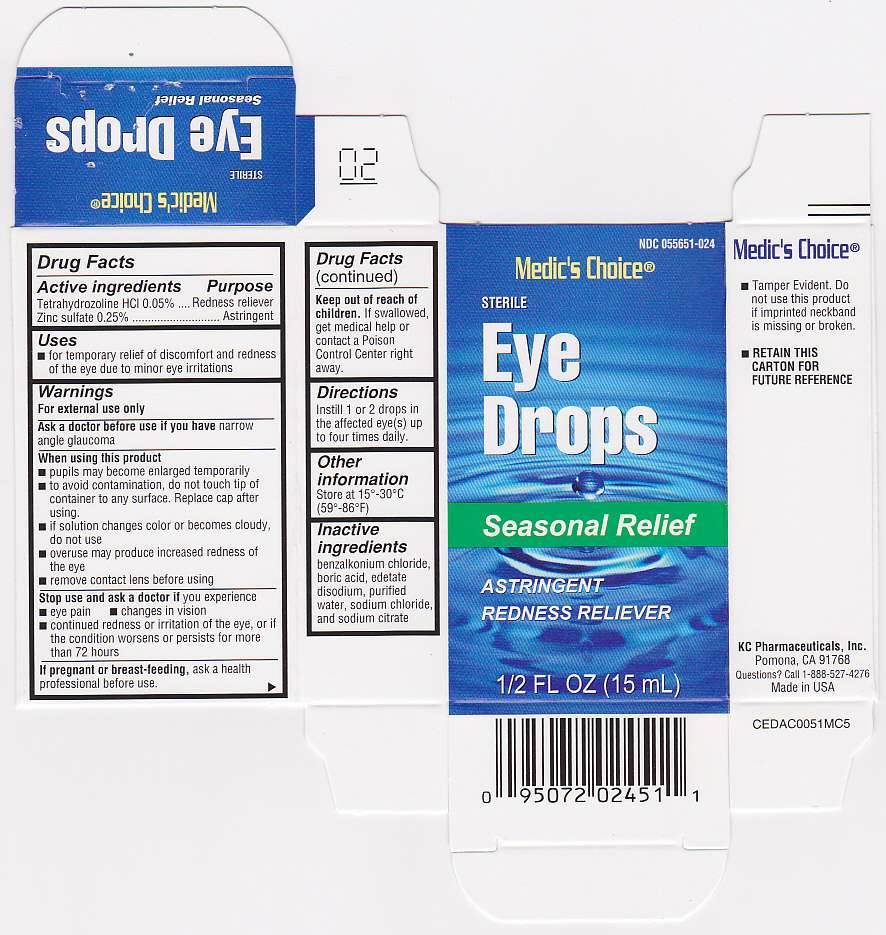
Eye Drops ACTetrahydrozoline hydrochloride, Zinc sulfate SOLUTION/ DROPS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||