DEXTROSE
Dextrose Injection, USP in VIAFLEX Plastic Container
FULL PRESCRIBING INFORMATION: CONTENTS*
- DEXTROSE DESCRIPTION
- CLINICAL PHARMACOLOGY
- DEXTROSE INDICATIONS AND USAGE
- DEXTROSE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- DEXTROSE ADVERSE REACTIONS
- DEXTROSE DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- DIRECTIONS FOR USE OF VIAFLEX PLASTIC CONTAINER
- PACKAGE LABELING - PRINCIPAL DISPLAY PANEL
FULL PRESCRIBING INFORMATION
DEXTROSE DESCRIPTION
Dextrose Injection, USP is a sterile, nonpyrogenic solution for fluid replenishment and caloric supply in single dose containers for intravenous administration. It contains no antimicrobial agents. Composition, osmolarity, pH, and caloric content are shown in Table 1.
| Size (mL) |
* Dextrose Hydrous, USP (g/L) |
Osmolarity (mOsmol/L) (calc.) |
pH | Caloric Content (kcal/L) |
|
| 5% Dextrose Injection, USP |
25 | 50 | 252 | 4.0 (3.2 to 6.5) |
170 |
| Quad pack | |||||
| 50 | |||||
| Single pack | |||||
| Quad pack | |||||
| Multi pack | |||||
| 100 | |||||
| Single pack | |||||
| Quad pack | |||||
| Multi pack | |||||
| 150 | |||||
| 250 | |||||
| 500 | |||||
| 1000 | |||||
| 10% Dextrose Injection, USP |
250 500 1000 |
100 | 505 | 4.0 (3.2 to 6.5) |
340 |
The VIAFLEX plastic container is fabricated from a specially formulated polyvinyl chloride (PL 146 Plastic). The amount of water that can permeate from inside the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the plastic container can leach out certain of its chemical components in very small amounts within the expiration period, e.g., di-2-ethylhexyl phthalate (DEHP), up to 5 parts per million. However, the safety of the plastic has been confirmed in tests in animals according to USP biological test for plastic containers as well as by tissue culture toxicity studies.
CLINICAL PHARMACOLOGY
Dextrose Injection, USP has value as a source of water and calories. It is capable of inducing diuresis depending on the clinical condition of the patient.
DEXTROSE INDICATIONS AND USAGE
Dextrose Injection, USP is indicated as a source of water and calories.
DEXTROSE CONTRAINDICATIONS
Solutions containing dextrose may be contraindicated in patients with known allergy to corn or corn products.
WARNINGS
Dextrose Injection, USP should not be administered simultaneously with blood through the same administration set because of the possibility of pseudoagglutination or hemolysis.
The intravenous administration of these solutions can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states, or pulmonary edema. The risk of dilutive states is inversely proportional to the electrolyte concentrations of the injections. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of the injections.
Excessive administration of dextrose injections may result in significant hypokalemia.
In very low birth weight infants, excessive or rapid administration of dextrose injection may result in increased serum osmolality and possible intracerebral hemorrhage.
PRECAUTIONS
Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Pressurizing intravenous solutions contained in flexible plastic containers to increase flow rates can result in air embolism if the residual air in the container is not fully evacuated prior to administration.
Use of a vented intravenous administration set with the vent in the open position could result in air embolism. Vented intravenous administration sets with the vent in the open position should not be used with flexible plastic containers.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations and acid base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
Dextrose Injection, USP should be used with caution in patients with overt or subclinical diabetes mellitus.
Pregnancy:
Teratogenic Effects
Pregnancy Category C.
Animal reproduction studies have not been conducted with Dextrose Injection, USP. It is also not known whether Dextrose Injection, USP can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Dextrose Injection, USP should be given to a pregnant woman only if clearly needed.
Pediatric Use
Dextrose is safe and effective for the stated indications in pediatric patients (see Indications and Usage).
Newborns – especially those born premature and with low birth weight - are at increased risk of developing hypo- or hyperglycemia and therefore need close monitoring during treatment with intravenous glucose solutions to ensure adequate glycemic control in order to avoid potential long term adverse effects. Hypoglycemia in the newborn can cause prolonged seizures, coma and brain damage. Hyperglycemia has been associated with intraventricular hemorrhage, late onset bacterial and fungal infection, retinopathy of prematurity, necrotizing enterocolitis, bronchopulmonary dysplasia, prolonged length of hospital stay, and death.
Geriatric Use
Clinical studies of Dextrose Injection, USP did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Do not administer unless solution is clear and seal is intact.
DEXTROSE ADVERSE REACTIONS
Hypersensitivity reactions, including anaphylaxis and chills.
Reactions which may occur because of the injection or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
DEXTROSE DOSAGE AND ADMINISTRATION
As directed by a physician. Dosage is dependent upon the age, weight and clinical condition of the patient as well as laboratory determinations.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
All injections in VIAFLEX plastic containers are intended for intravenous administration using sterile equipment.
The dosage selection and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low birth weight infants, because of the increased risk of hyperglycemia/ hypoglycemia. Frequent monitoring of serum glucose concentrations is required when dextrose is prescribed to pediatric patients, particularly neonates and low birth weight infants. The infusion rate and volume depends on the age, weight, clinical and metabolic conditions of the patient, concomitant therapy and should be determined by the consulting physician experienced in pediatric intravenous fluid therapy.
Additives may be incompatible. Complete information is not available.
Those additives known to be incompatible should not be used. Consult with pharmacist, if available. If, in the informed judgment of the physician, it is deemed advisable to introduce additives, use aseptic technique. Mix thoroughly when additives have been introduced. Do not store solutions containing additives.
HOW SUPPLIED
Dextrose Injection, USP in VIAFLEX plastic container is available as follows:
| Code |
Size (mL) |
NDC | Product Name |
| 25 | |||
| 2B0080 | Quad pack | 0338-0017-10 | 5% Dextrose Injection, USP |
| 50 | |||
| 2B0086 | Single pack | 0338-0017-41 | 5% Dextrose Injection, USP |
| 2B0081 | Quad pack | 0338-0017-11 | |
| 2B0088 | Multi pack | 0338-0017-31 | |
| 100 | |||
| 2B0087 | Single pack | 0338-0017-48 | 5% Dextrose Injection, USP |
| 2B0082 | Quad pack | 0338-0017-18 | |
| 2B0089 | Multi pack | 0338-0017-38 | |
| 2B0061 | 150 | 0338-0017-01 | |
| 2B0062 | 250 | 0338-0017-02 | |
| 2B0063 | 500 | 0338-0017-03 | |
| 2B0064 | 1000 | 0338-0017-04 | |
| 2B0162 | 250 | 0338-0023-02 | 10% Dextrose Injection, USP |
| 2B0163 | 500 | 0338-0023-03 | |
| 2B0164 | 1000 | 0338-0023-04 |
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. It is recommended the product be stored at room temperature (25°C/77°F); brief exposure up to 40°C/104°F does not adversely affect the product.
DIRECTIONS FOR USE OF VIAFLEX PLASTIC CONTAINER
For Information on Risk of Air Embolism - see Precautions
To Open
Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired. If supplemental medication is desired, follow “To Add Medication” directions below.
Preparation for Administration
- Suspend container from eyelet support.
- Remove protector from outlet port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
To Add Medication
Warning: Additives may be incompatible.
To add medication before solution administration
- Prepare medication site.
- Using syringe with 19 to 22 gauge needle, puncture resealable medication port and inject.
- Mix solution and medication thoroughly. For high density medication such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
To add medication during solution administration
- Close clamp on the set.
- Prepare medication site.
- Using syringe with 19 to 22 gauge needle, puncture resealable medication port and inject.
- Remove container from IV pole and/or turn to an upright position.
- Evacuate both ports by squeezing them while container is in the upright position.
- Mix solution and medication thoroughly.
- Return container to in-use position and continue administration.
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Printed in USA
07-19-69-268
Rev. November 2013
Baxter, PL 146, and Viaflex are trademarks of Baxter International Inc.
PACKAGE LABELING - PRINCIPAL DISPLAY PANEL

2B0063Q
24-500 ML
VIAFLEX(R) CONTAINER
10% DEXTROSE INJECTION, USP
SECONDARY BAR CODE
(17) YYMMOO (10) XXXXX
PRIMARY BAR CODE
(01) 50303380023030
EXP
XXXXX
LOT
XXXXX
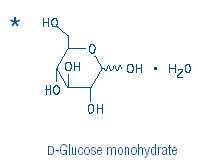
DEXTROSEDEXTROSE MONOHYDRATE INJECTION, SOLUTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DEXTROSEDEXTROSE MONOHYDRATE INJECTION, SOLUTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||