Citalopram Hydrobromide
FULL PRESCRIBING INFORMATION: CONTENTS*
- BOXED WARNING
- CITALOPRAM HYDROBROMIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACODYNAMICS
- PHARMACOKINETICS
- INDICATIONS & USAGE
- CITALOPRAM HYDROBROMIDE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- LABORATORY TESTS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- LABOR & DELIVERY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- CITALOPRAM HYDROBROMIDE ADVERSE REACTIONS
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- ANIMAL PHARMACOLOGY & OR TOXICOLOGY
- SPL MEDGUIDE
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
BOXED WARNING
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of Citalopram HBr or any other antidpressant in child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in teh risk of suicidalit with antidpressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidpressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers shouldbe advised of the need for close observation and communication with the prescriber. Citalopram HBr is not approved for use in pediatric patients. (See WARNINGS: Clinical Worsening and Suicide RiskPRECAUTIONS: Information for PatientsPRECAUTIONS: Pediatric UseCITALOPRAM HYDROBROMIDE DESCRIPTION
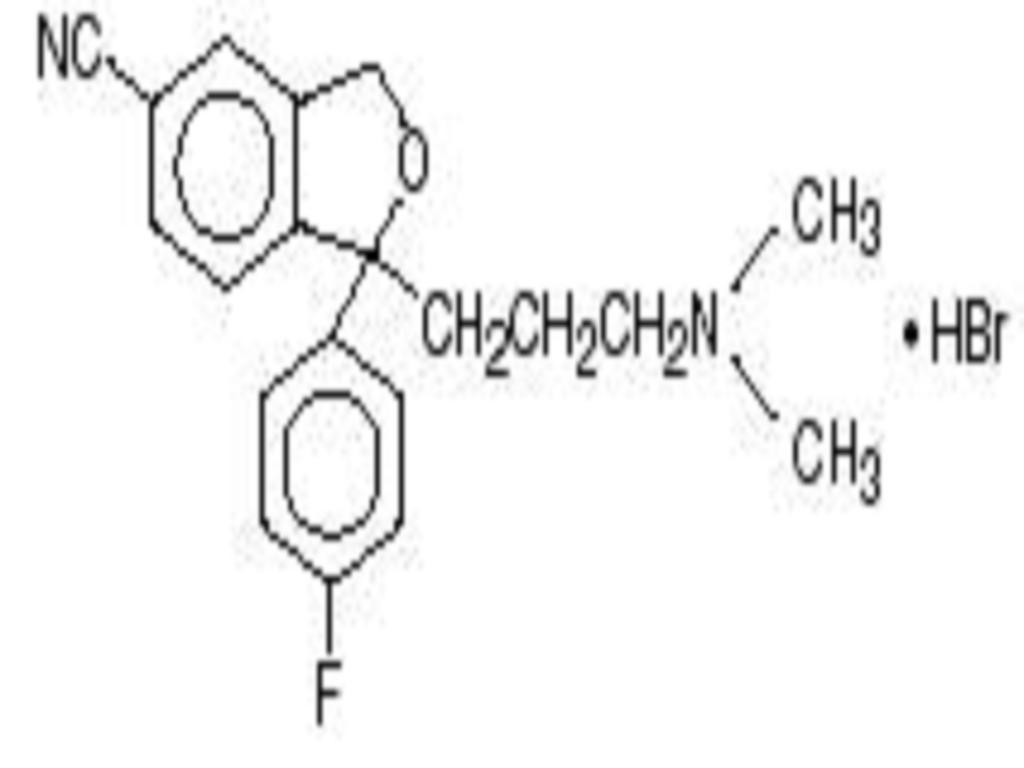
CLINICAL PHARMACOLOGY
PHARMACODYNAMICS
PHARMACOKINETICS
DOSAGE AND ADMINISTRATION
DOSAGE AND ADMINISTRATION
Drug InteractionsPRECAUTIONS
Clinical Efficacy Trials
INDICATIONS & USAGE
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY
CITALOPRAM HYDROBROMIDE CONTRAINDICATIONS
WARNINGSPRECAUTIONS
WARNINGS
Clinical Worsening and Suicide RiskPRECAUTIONSDOSAGE AND ADMINISTRATIONDiscontinuation of Treatment with Citalopram
Potential for Interaction with Monoamine Oxidase Inhibitors
Serotonin Syndrome or Neuroleptic Malignant Syndrome (NMS)-like Reactions
PRECAUTIONS
GeneralDOSAGE AND ADMINISTRATION
Geriatric Use
DOSAGE AND ADMINISTRATION
DOSAGE AND ADMINISTRATION
INFORMATION FOR PATIENTS
LABORATORY TESTS
DRUG INTERACTIONS
WARNINGS-Serotonin SyndromePRECAUTIONS - Drug InteractionsWARNINGS - Serotonin Syndrome
CONTRAINDICATIONSWARNINGS.
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
Pregnancy-Nonteratogenic Effects
WARNINGS
DOSAGE AND ADMINISTRATION
LABOR & DELIVERY
NURSING MOTHERS
PEDIATRIC USE
BOX WARNINGWARNINGSClinical Worsening and Suicide RiskGERIATRIC USE
DOSAGE AND ADMINISTRATIONPRECAUTIONS, Hyponatremia
CLINICAL PHARMACOLOGY
DOSAGE AND ADMINISTRATION
CITALOPRAM HYDROBROMIDE ADVERSE REACTIONS
Adverse Findings Observed in Short-Term, Placebo-Controlled Trials
TABLE 3
Other Events Observed During the Premarketing Evaluation of Citalopram HBr
Other Events Observed During the Postmarketing Evaluation of Citalopram HBr
DRUG ABUSE AND DEPENDENCE
Controlled Substance ClassPhysical and Psychological Dependence
OVERDOSAGE
Human ExperienceManagement of Overdose
DOSAGE & ADMINISTRATION
Initial TreatmentSpecial Populations
Treatment of Pregnant Women During the Third Trimester
PRECAUTIONS
Maintenance Treatment
Clinical TrialsCLINICAL PHARMACOLOGY
Discontinuation of Treatment with Citalopram
PRECAUTIONS
Switching Patients To or From a Monoamine Oxidase Inhibitor
CONTRAINDICATIONSWARNINGS
HOW SUPPLIED
ANIMAL PHARMACOLOGY & OR TOXICOLOGY
Retinal Changes in RatsCardiovascular Changes in Dogs
SPL MEDGUIDE
-
● all risks and benefits of treatment with antidepressant medicines
-
● all treatment choices for depression or other serious mental illness
-
● Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
-
● Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
-
● Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
-
● thoughts about suicide or dying
-
● attempts to commit suicide
-
● new or worse depression
-
● new or worse anxiety
-
● feeling very agitated or restless
-
● panic attacks
-
● trouble sleeping (insomnia)
-
● new or worse irritability
-
● acting aggressive, being angry, or violent
-
● acting on dangerous impulses
-
● an extreme increase in activity and talking (mania)
-
● other unusual changes in behavior or mood
-
● Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
-
● Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
-
● Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
-
● Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
-
● Not all antidepressant medicines prescribed for children are FDA approved for use in children. Talk to your child's healthcare provider for more information.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

Citalopram HydrobromideCitalopram Hydrobromide TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!