XEROSTOM
Xerostom Mouthwash
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredient
- Purpose Section
- Keep out of reach of children
- Indications & Usage Section
- Warnings
- Dosage and administration
- Inactive Ingredient
- Principal Display panel
FULL PRESCRIBING INFORMATION
Active Ingredient
Sodium Fluoride 0.055%
Purpose Section
Anticavity Rinse
Keep out of reach of children
Keep out of reach of children 12yrs of age and under
Indications & Usage Section
Aids in the prevention of dental cavities.
Warnings
Keep out of reach of children 12 years of age and under.
For external use only. If you accidentally swallow more than used for rinsing, get medical help or contact a Poisson Control Center immediately.
Do not use if you are allergic to any of the ingredients.
Dosage and administration
Adults and children over 12 years use at least twice a day after brushing.
Pour 10 mililiters; vigorously swish 10mililiters of rinse between teeth for I minute.
Then spit out.
Inactive Ingredient
Water
Betaine
Glycerin
Peg-40 Hydrogenated Castor Oil
Propylene Glycol
Xylitol
Aroma
Potassium Phosphate
Diazolidinyl Urea
Allantoin
Olive oil
Sodium Methyl paraben
Sodium Propyl Paraben
Chlorophyll
D-Panthenol
Tocopheryl Acetate
Sucralose
Parsley Oil.
Principal Display panel
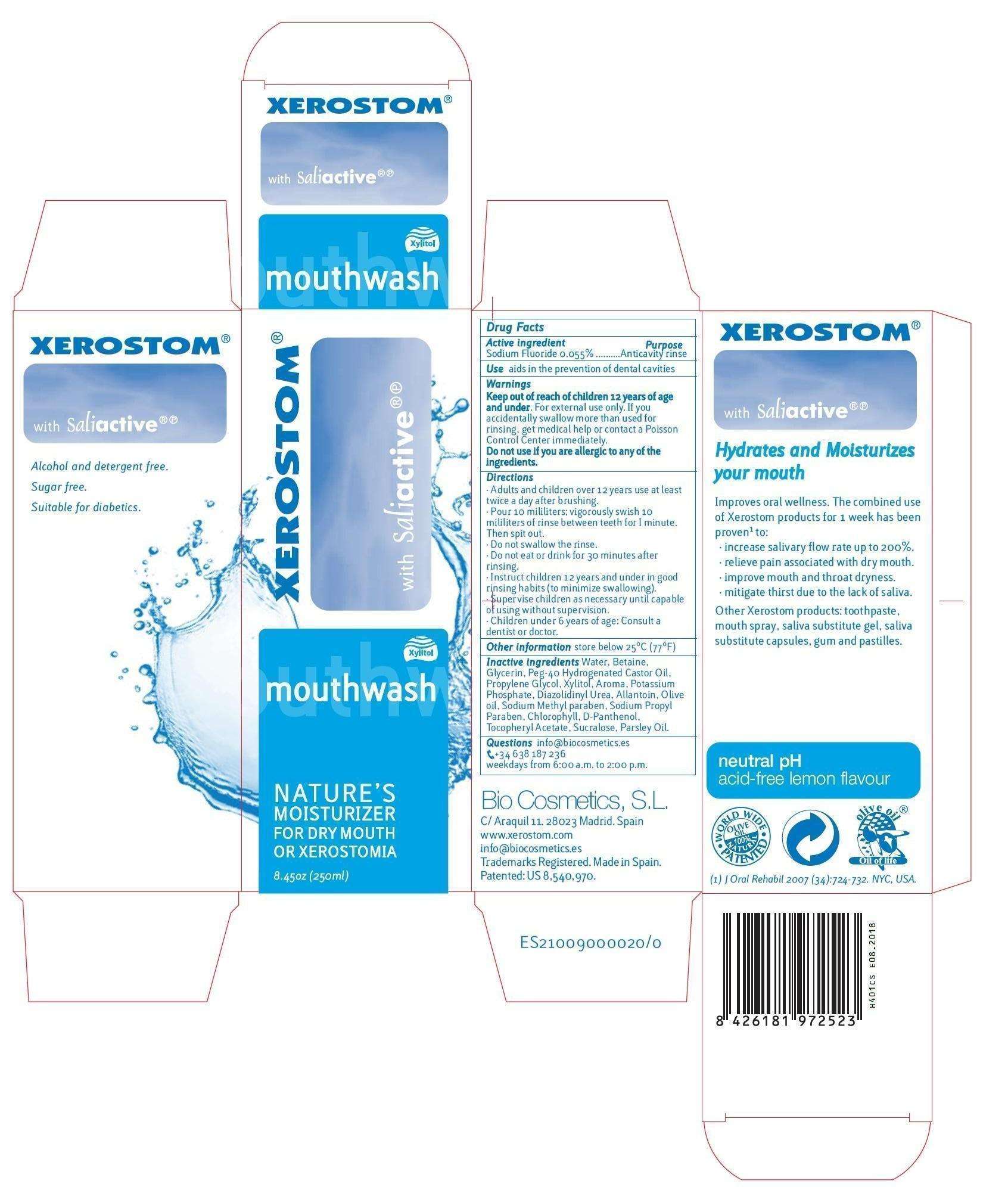
XEROSTOMSODIUM FLUORIDE MOUTHWASH
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||