TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor
TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor
FULL PRESCRIBING INFORMATION: CONTENTS*
- TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor
- Active ingredients (in each 5 mL teaspoonful)
- TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor Uses
- Warnings
- Directions
- TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor Other information
- Inactive ingredients
- Questions?
- TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor 4 fl oz/118ml (58238-227-04)
FULL PRESCRIBING INFORMATION
TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor
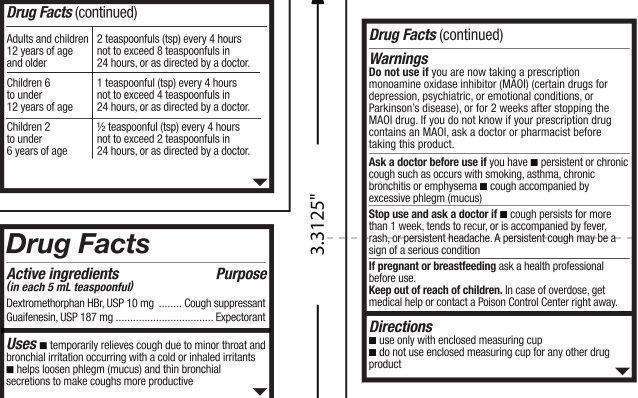
Active ingredients (in each 5 mL teaspoonful)
Dextromethorphan HBr, USP 10 mg
Guaifenesin, USP 187 mg
Purpose
Cough suppressant
Expectorant
TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor Uses
- temporarily relieves cough due to minor throat and bronchial irritation occurring with a cold or inhaled irritants
- helps loosen phlegm (mucus) and thin bronchial secretions to make coughs more productive
Warnings
Do not use if
you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if
you have
- persistent or chronic cough such as occurs with asthma
- cough accompanied by excessive phlegm (mucus)
Stop use and ask a doctor if
- cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache. A persistent cough may be a sign of a serious condition.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
■ use only with enclosed measuring cup
■ do not use enclosed measuring cup for any other drug product
Adults and children 12 years of age and older: 2 teaspoonfuls (tsp) every 6 hours not to exceed 8 teaspoonfuls in 24 hours, or
as directed by a doctor.
Children 6 to under 12 years of age: 1 teaspoonful (tsp) every 6 hours not to exceed 4 teaspoonfuls in 24 hours, or
as directed by a doctor.
Children 2 to under 6 years of age: ½ teaspoonful (tsp) every 4 hours not to exceed 2 teaspoonfuls in 24 hours, or
as directed by a doctor.
TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor Other information
- Store at room temperature 15°C-30°C (59°F-86°F)
Inactive ingredients
cherry flavor, citric acid, glycerin, methylparaben, monoammonium glycyrrhizinate, potassium citrate, potassium sorbate, propylparaben, propylene glycol, purified water, raspberry flavor, sorbitol, sucralose
Questions?
Call 1-787-701-3312. You may also report serious side effects to this phone number.
TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY Flavor 4 fl oz/118ml (58238-227-04)

TRISPEC DMX Cough Suppressant Expectorant CHERRY RASPBERRY FlavorDEXTROMETHORPHAN HYDROBROMIDE, GUAIFENESIN LIQUID
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||