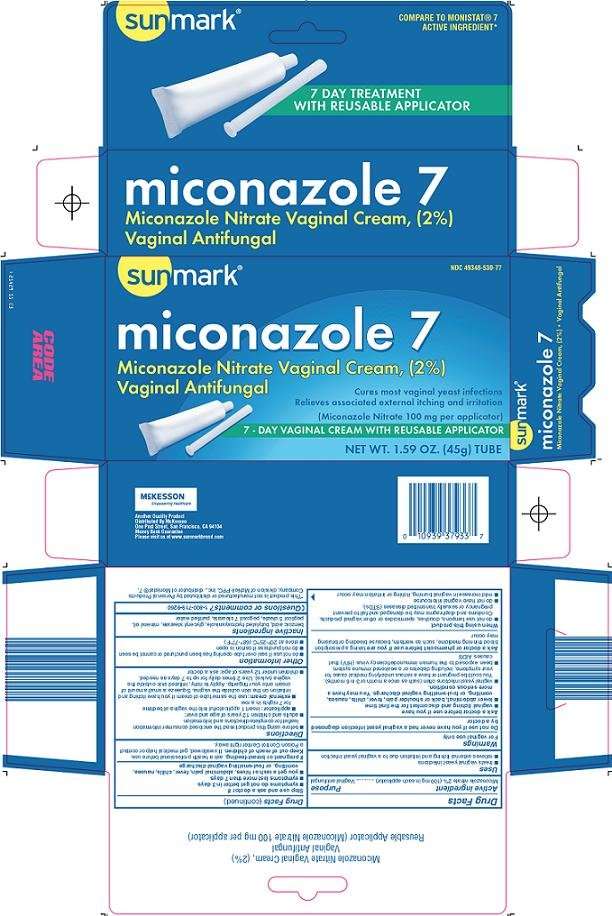
Sunmark Miconazole 7
McKesson Miconazole 7 Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient
- Purpose
- Sunmark Miconazole 7 Uses
- Warnings
- Directions
- Sunmark Miconazole 7 Other information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Active ingredient
Miconazole nitrate 2% (100 mg in each applicator)
Purpose
Vaginal antifungal
Sunmark Miconazole 7 Uses
- treats vaginal yeast infections
- relieves external itching and irritation due to a vaginal yeast infection
Warnings
For vaginal use only
Do not use
if you have never had a vaginal yeast infection diagnosed by a doctor
Ask a doctor before use if you have
- vaginal itching and discomfort for the first time
- lower abdominal, back or shoulder pain, fever, chills, nausea, vomiting, or foul-smelling vaginal discharge. You may have a more serious condition.
- vaginal yeast infections often (such as once a month or 3 in 6 months). You could be pregnant or have a serious underlying medical cause for your symptoms, including diabetes or a weakened immune system.
- been exposed to the human immunodeficiency virus (HIV) that causes AIDS
Ask a doctor or pharmacist before use if you are
taking a prescription blood thinning medicine, such as warfarin, because bleeding or bruising may occur
When using this product
- do not use tampons, douches, spermicides or other vaginal products. Condoms and diaphragms may be damaged and fail to prevent pregnancy or sexually transmitted diseases (STDs).
- do not have vaginal intercourse
- mild increase in vaginal burning, itching or irritation may occur
Stop use and ask a doctor if
- symptoms do not get better in 3 days
- symptoms last more than 7 days
- you get a rash or hives, abdominal pain, fever, chills, nausea, vomiting, or foul-smelling vaginal discharge
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- before using this product read the enclosed consumer information leaflet for complete directions and information
- adults and children 12 years of age and over:
- applicator: insert 1 applicatorful into the vagina at bedtime for 7 nights in a row
- external cream: use the same tube of cream if you have itching and irritation on the skin outside the vagina. Squeeze a small amount of cream onto your fingertip. Apply to itchy, irritated skin outside the vagina (vulva). Use 2 times daily for up to 7 days as needed.
- children under 12 years of age: ask a doctor
Sunmark Miconazole 7 Other information
- do not use if seal over tube opening has been punctured or cannot be seen
- do not purchase if carton is open
- store at 20°-25°C (68°-77°F)
Inactive ingredients
benzoic acid, butylated hydroxyanisole, glyceryl stearate, mineral oil, peglicol 5 oleate, pegoxol 7 stearate, purified water
Questions or comments?
1-800-719-9260
Principal Display Panel
Compare to Monistat® 7 Active Ingredient
7 Day Treatment with Reusable Applicator
Miconazole 7
Miconazole Nitrate Vaginal Cream, (2%)
Vaginal Antifungal
Cures Most Vaginal Yeast Infections
Relieves Associated External Itching and Irritation
(Miconazole Nitrate 100 mg per applicator)
7-Day Vaginal Cream with Reusable Applicator
Sunmark Miconazole 7Miconazole nitrate CREAM
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||