Raggedy Ann and Andy
Raggedy Ann&Andy Tropical Breeze ANTIBACTERIAL HAND SOAP
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredient
- Purpose
- Raggedy Ann and Andy Uses
- Warnings
- Directions
- Inactive Ingredients
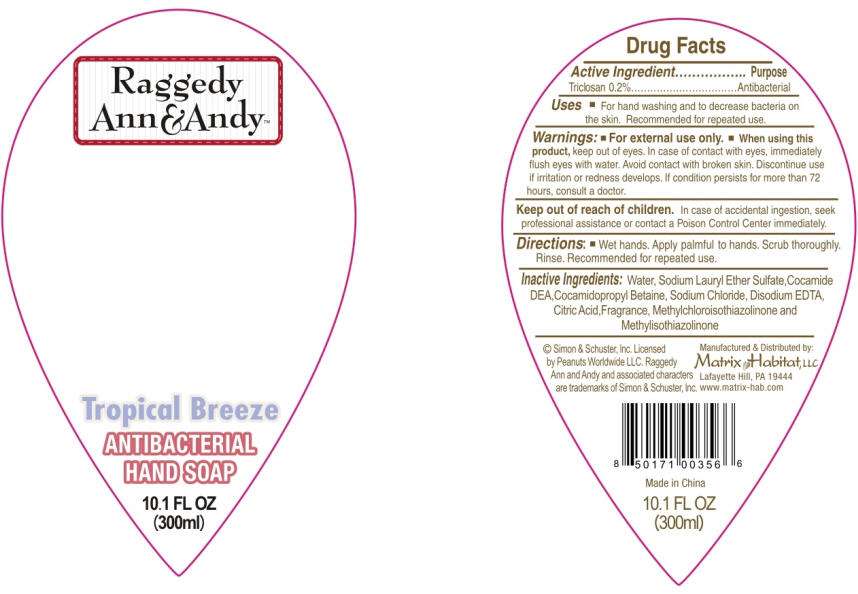
- PRINCIPAL DISPLAY PANEL - 300ml Bottle Label
FULL PRESCRIBING INFORMATION
Drug Facts
Active Ingredient
Triclosan 0.2%
Purpose
Antibacterial
Raggedy Ann and Andy Uses
- For hand washing and to decrease bacteria on the skin. Recommended for repeated use.
Warnings
- For external use only.
- When using this product, keep out of eyes. In case of contact with eyes, immediately flush eyes with water. Avoid contact with broken skin. Discontinue use if irritation or redness develops. If condition persists for more than 72 hours, consult a doctor.
Keep out of reach of children. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Directions
- Wet hands. Apply palmful to hands. Scrub thoroughly. Rinse. Recommended for repeated use.
Inactive Ingredients
Water, Sodium Lauryl Ether Sulfate,Cocamide DEA,Cocamidopropyl Betaine, Sodium Chloride, Disodium EDTA, Citric Acid,Fragrance, Methylchloroisothiazolinone and Methylisothiazolinone
PRINCIPAL DISPLAY PANEL - 300ml Bottle Label
Raggedy
Ann&Andy™
Tropical Breeze
ANTIBACTERIAL
HAND SOAP
10.1 FL OZ
(300ml)
Raggedy Ann and AndyTriclosan SOAP
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!