Mirtazapine
FULL PRESCRIBING INFORMATION: CONTENTS*
- BOXED WARNING
- MIRTAZAPINE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- MIRTAZAPINE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- LABORATORY TESTS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- MIRTAZAPINE ADVERSE REACTIONS
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- SPL MEDGUIDE
- INACTIVE INGREDIENT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
BOXED WARNING
Suicidality and Antidepressant Drugs
Warnings: Clinical Worsening and Suicide RiskPrecautions: Information for PatientsPrecautions: Pediatric Use
MIRTAZAPINE DESCRIPTION
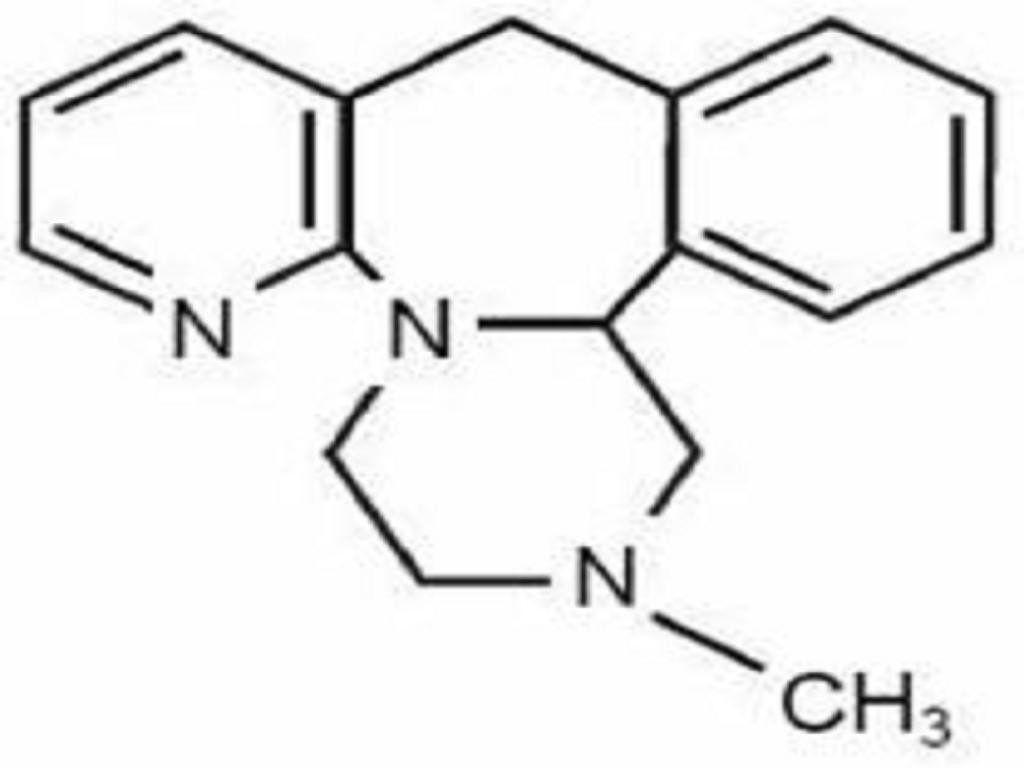
CLINICAL PHARMACOLOGY
PharmacodynamicsPharmacokinetics
Special Populations
PRECAUTIONSDOSAGE AND ADMINISTRATION
PRECAUTIONS
Pharmacokinetics
PRECAUTIONSDOSAGE AND ADMINISTRATION
PRECAUTIONSDOSAGE AND ADMINISTRATION
Clinical Trials Showing Effectiveness
INDICATIONS & USAGE
CLINICAL PHARMACOLOGYCLINICAL PHARMACOLOGY
MIRTAZAPINE CONTRAINDICATIONS
CONTRAINDICATIONSWARNINGS
Clinical Worsening and Suicide RiskAgranulocytosis
MAO Inhibitors
PRECAUTIONS
GeneralSomnolence
Information for Patients
Dizziness
Increased Appetite/Weight Gain
PRECAUTIONS: Pediatric Use
Cholesterol/Triglycerides
Transaminase Elevations
CLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
Activation of Mania/Hypomania
Seizure
Use in Patients with Concomitant Illness
CLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
INFORMATION FOR PATIENTS
Information for PatientsLABORATORY TESTS
Laboratory TestsDRUG INTERACTIONS
Drug InteractionsCLINICAL PHARMACOLOGY
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
NURSING MOTHERS
Nursing MothersPEDIATRIC USE
Pediatric UseBOX WARNINGWARNINGSClinical Worsening and Suicide Risk
PRECAUTIONSIncreased Appetite/Weight Gain
GERIATRIC USE
Geriatric UseCLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
MIRTAZAPINE ADVERSE REACTIONS
Associated with Discontinuation of TreatmentCommon Adverse Events Associated With Discontinuation of Treatment in 6-week U.S. Mirtazapine Trials Percentage of Patients Discontinuing with Adverse Event
Commonly Observed Adverse Events in U.S. Controlled Clinical Trials
Common Treatment-Emergent Adverse Events Associated with the Use of Mirtazapine in 6-week U.S. Trials Percentage of Patients Reporting Adverse Event
Adverse Events Occurring at an Incidence of 1% or More Among Mirtazapine Treated Patients
INCIDENCE OF ADVERSE CLINICAL EXPERIENCES (IN SHORT-TERM U.S.CONTROLLED STUDIES1Body as a WholeDigestive SystemMetabolic and Nurtritional DisordersMusculoskeletal SystemNervous SystemRespiratory SystemUrogenital System
ECG Changes
Other Adverse Events Observed During the Premarketing Evaluation of Mirtazapine
WARNINGSPRECAUTIONS
Other Adverse Events Observed During Postmarketing Evaluation of Mirtazapine
DRUG ABUSE AND DEPENDENCE
DRUG ABUSE AND DEPENDENCEControlled Substance Class
Physical and Psychological Dependence
OVERDOSAGE
OVERDOSAGEHuman Experience
Overdose Management
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATIONInitial Treatment
Elderly and Patients with Renal or Hepatic Impairment
PRECAUTIONSCLINICAL PHARMACOLOGY
Maintenance/Extended Treatment
CLINICAL PHARMACOLOGY
Switching Patients To or From a Monoamine Oxidase Inhibitor
HOW SUPPLIED
SPL MEDGUIDE
MEDICATION GUIDE-
● all risks and benefits of treatment with antidepressant medicines
-
● all treatment choices for depression or other serious mental illness
-
● Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
-
● Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
-
● Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
-
● thoughts about suicide or dying
-
● attempts to commit suicide
-
● new or worse depression
-
● new or worse anxiety
-
● feeling very agitated or restless
-
● panic attacks
-
● trouble sleeping (insomnia)
-
● new or worse irritability
-
● acting aggressive, being angry, or violent
-
● acting on dangerous impulses
-
● an extreme increase in activity and talking (mania)
-
● other unusual changes in behavior or mood
-
● Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
-
● Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
-
● Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
-
● Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
-
● Not all antidepressant medicines prescribed for children are FDA approved for use in children. Talk to your child's healthcare provider for more information.
INACTIVE INGREDIENT
INACTIVE INGREDIENTS:ANHYDROUS LACTOSE
COLLOIDAL SILICON DIOXIDE
MAGNESIUM STEARATE
CELLULOSE, MICROCRYSTALLINE
HYPROMELLOSE 2910 (3 MPA.S)
HYPROMELLOSE 2910 (6 MPA.S)
POLYETHYLENE GLYCOL
POLYSORBATE 80
TITANIUM DIOXIDE
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

MirtazapineMirtazapine TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!