GUNA-GERIATRICS
DRUG FACTS
FULL PRESCRIBING INFORMATION: CONTENTS*
FULL PRESCRIBING INFORMATION
ACTIVE INGREDIENTS/PURPOSE
ACTH 6X ADRENAL SUPPORT
ARNICA MONTANA 6X ANTI-INFLAMMATORY
BARIUM OXALOSUCCINATE 3X FORGETFULNESS
BARYTA CARBONICA 6X IMPROVES MENTAL ATTENTION
BRAIN DERIVED NEUROTROPHIC FACTOR 4C HELPS MENTAL ACTIVITY
DL-MALIC ACID 3X PROMOTE CELL METABOLISM
FRONTAL LOBE 6X STIMULATES MENTAL ALERTNESS
GLANDULA SUPRARENALIS 6X DETOXIFICATION
HEPAR 6X DETOXIFICATION
HYPOTHALAMUS 6X IMPROVES MENTAL ATTENTION
INSULINE-LIKE GROWTH FACTOR-1 4C ENHANCES PROTEIN ANABOLISM
LUTEINIZING HORMONE-RELEASING HORMONE 6X HELPS MANAGE STRESS
MELATONIN 4C HELPS MANAGE STRESS
NEUROTROPHIN 3 4C HELPS MENTAL ACTIVITY
NEUROTROPHIN 4 4C HELPS MENTAL ACTIVITY
OXYTOCIN 6X STIMULATES SOCIAL RECOGNITION
PARABENZOCHINON 3X ANTIOXIDANT
PHENYLALANINE 3X STIMULATES MENTAL ALERTNESS
PLUMBUM METALLICUM 6X ANTIAGING
PYRUVIC ACID 3X ANTIOXIDANT
THYROTROPIN-RELEASING HORMONE 6X HELPS MANAGE STRESS
THYROXINE T4 6X STIMULATES MENTAL ALERTNESS
USES
For the temporary relief of cognitive symptoms due to aging: poor memory, impaired concentration
WARNINGS
Stop use and ask doctor if symptoms of poor memory, impaired concentration worsen or persist more than 5 days
PREGNANCY
If pregnant or breast-feeding ask a doctor before use
WARNINGS
Keep this and all medicines out of reach of children
DIRECTIONS
Take 15 minutes before meals
Adults and children 12 years and older 20 drops in a little water, 2 times per day
Children between 12 years and 6 years of age 10 drops in a little water, 2 times per day
Children under 6 years Ask a Physician
QUESTIONS
Questions?: info@gunainc.com, tel. (484) 223-3500
PRINCIPAL DISPLAY PANEL
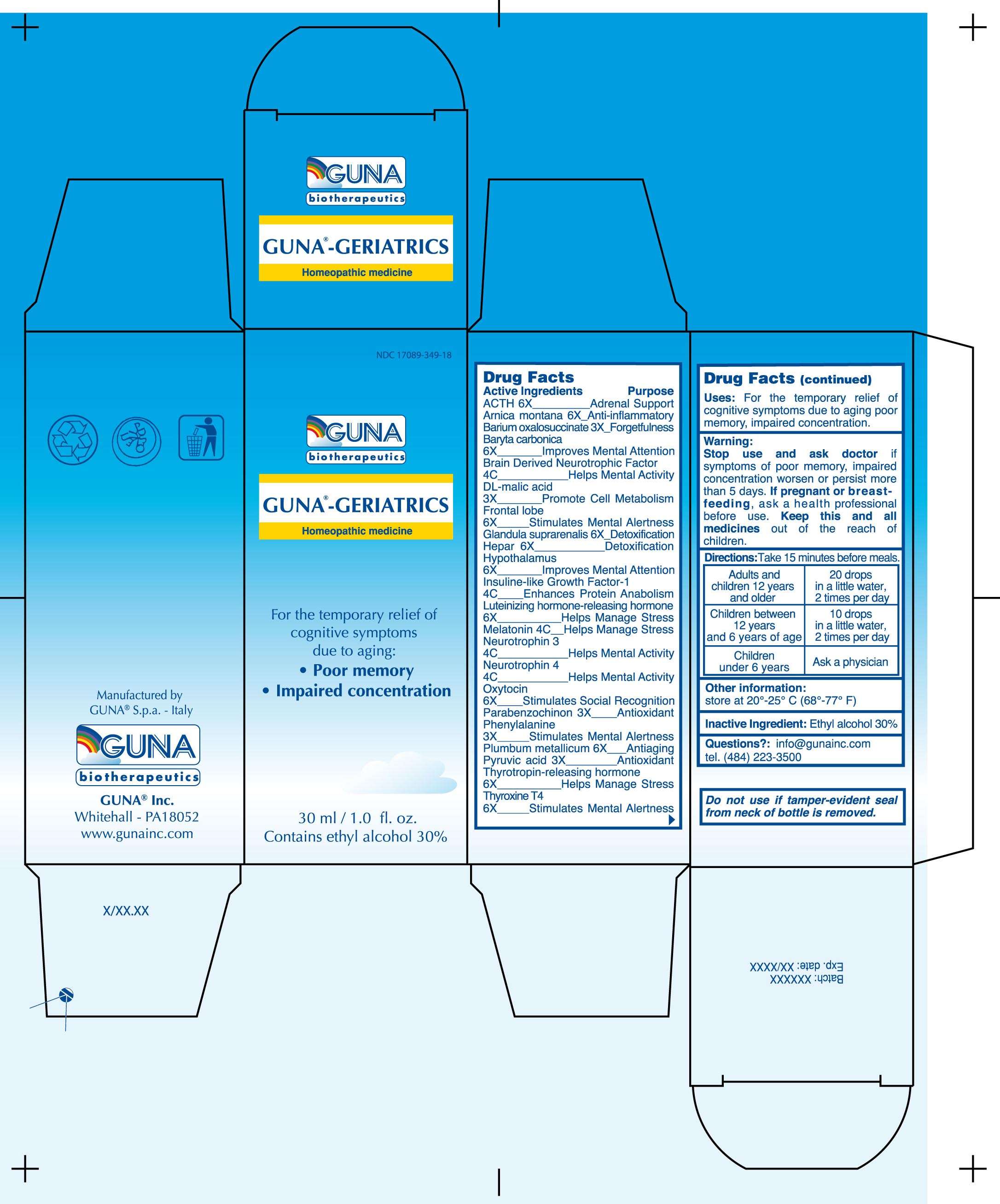
GUNA-GERIATRICS1,4-BENZOQUINONE - ARNICA MONTANA - OXYTOCIN - BARIUM CARBONATE - BARIUM OXALOSUCCINATE - CORTICOTROPIN - LEAD - LEVOTHYROXINE - LUTRELIN - MALIC ACID - MELATONIN - NEUROTROPHIN-3 - NEUROTROPHIN-4 - PHENYLALANINE - PORK LIVER - PYRUVIC ACID - RINFABATE - SUS SCROFA ADRENAL GLAND - SUS SCROFA FRONTAL LOBE - SUS SCROFA HYPOTHALAMUS - THYROTROPIN ALFA - BRAIN-DERIVED NEUROTROPHIC FACTOR HUMAN - SOLUTION/ DROPS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||