Claritin-D
Claritin-D 24 Hour
FULL PRESCRIBING INFORMATION: CONTENTS*
- Claritin-D Uses
- Warnings
- Directions
- Claritin-D Other information
- Inactive ingredients
- Questions or comments?
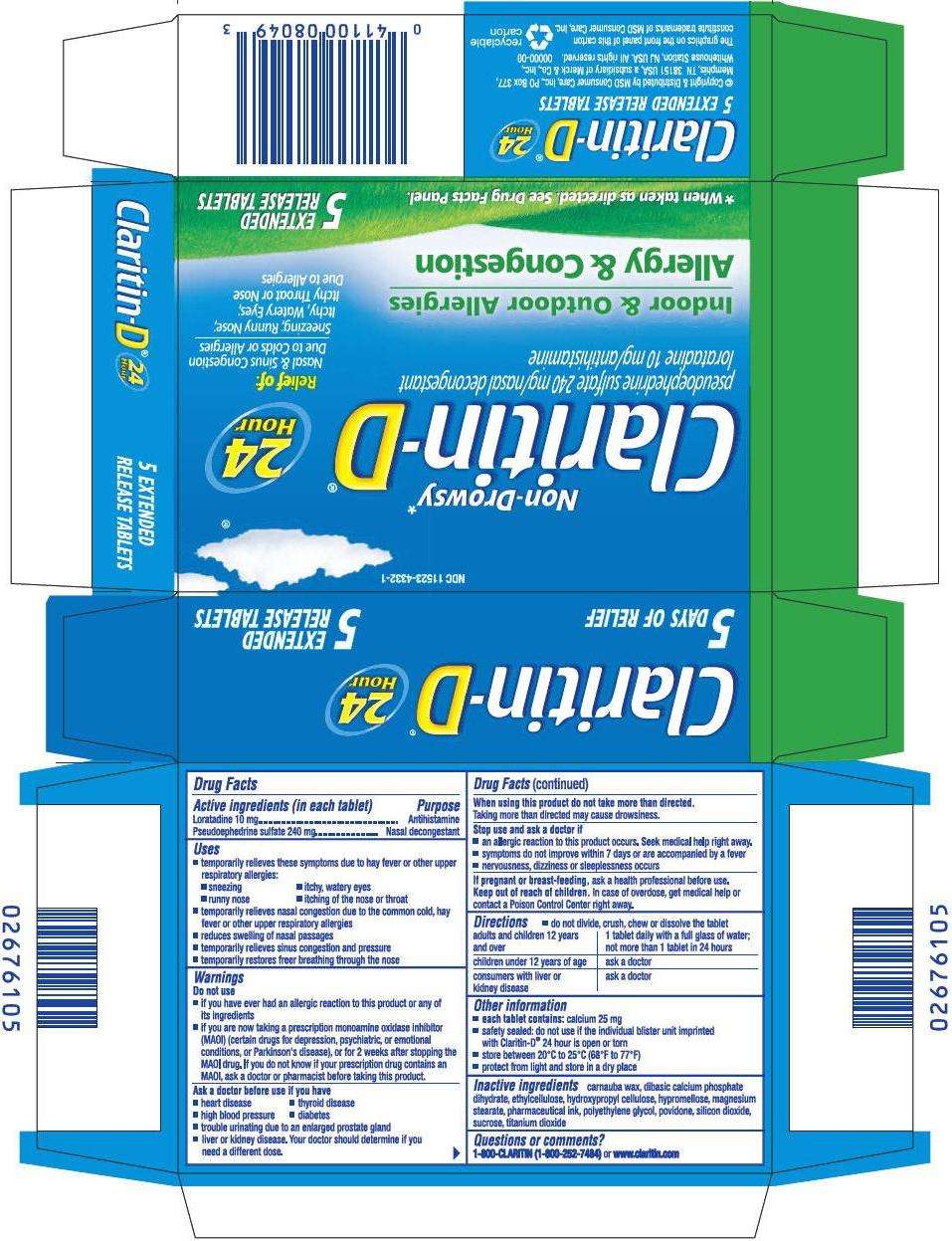
- PRINCIPAL DISPLAY PANEL - 5 Tablet Blister Pack Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
Purpose
| Active ingredient (in each tablet) | Purpose |
|---|---|
| Loratadine 10 mg | Antihistamine |
| Pseudoephedrine sulfate 240 mg | Nasal decongestant |
Claritin-D Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- sneezing
- itchy, watery eyes
- runny nose
- itching of the nose or throat
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily restores freer breathing through the nose
Warnings
Do not use
- if you have ever had an allergic reaction to this product or any of its ingredients
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- heart disease
- thyroid disease
- high blood pressure
- diabetes
- trouble urinating due to an enlarged prostate gland
- liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Stop use and ask a doctor if
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve within 7 days or are accompanied by a fever
- nervousness, dizziness or sleeplessness occurs
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- do not divide, crush, chew or dissolve the tablet
| adults and children 12 years and over | 1 tablet daily with a full glass of water; not more than 1 tablet in 24 hours |
| children under 12 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Claritin-D Other information
- each tablet contains: calcium 25 mg
- safety sealed: do not use if the individual blister unit imprinted with Claritin-D® 24 hour is open or torn
- store between 20° to 25°C (68° to 77°F)
- protect from light and store in a dry place
Inactive ingredients
carnauba wax, dibasic calcium phosphate dihydrate, ethylcellulose, hydroxypropyl cellulose, hypromellose, magnesium stearate, pharmaceutical ink, polyethylene glycol, povidone, silicon dioxide, sucrose, titanium dioxide
Questions or comments?
1-800-CLARITIN (1-800-252-7484) or www.claritin.com
Distributed by MSD Consumer Care, Inc., PO Box 377,
Memphis, TN 38151 USA, a subsidiary of Merck & Co., Inc.,
Whitehouse Station, NJ USA.
PRINCIPAL DISPLAY PANEL - 5 Tablet Blister Pack Carton
NDC 11523-4332-1
Non-Drowsy*
Claritin-D
®
pseudoephedrine sulfate 240 mg/nasal decongestant
loratadine 10 mg/antihistamine
Indoor & Outdoor Allergies
Allergy & Congestion
24
Hour
Relief of:
Nasal & Sinus Congestion
Due to Colds or Allergies
Sneezing; Runny Nose;
Itchy, Watery Eyes;
Itchy Throat or Nose
Due to Allergies
*When taken as directed. See Drug Facts Panel.
5
EXTENDED
RELEASE TABLETS
Claritin-DLoratadine and pseudoephedrine sulfate TABLET, EXTENDED RELEASE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||