Home – Chelidonium Homaccord
Chelidonium Homaccord
Heel Inc
Chelidonium-Homaccord 1.1ml Oral Vial
FULL PRESCRIBING INFORMATION: CONTENTS*
FULL PRESCRIBING INFORMATION
ACTIVE INGREDIENTS
Active ingredients:PURPOSE
KEEP OUT OF REACH OF CHILDREN
Keep this and all medicines out of the reach of children.CHELIDONIUM HOMACCORD INDICATIONS AND USAGE
Indications:
- Bloating
- Spasms
- Colic and flatulence
- Constipation or diarrhea
WARNINGS
Warnings: CHELIDONIUM HOMACCORD DOSAGE AND ADMINISTRATION
Dosasge:INACTIVE INGREDIENTS
Inactive ingredients: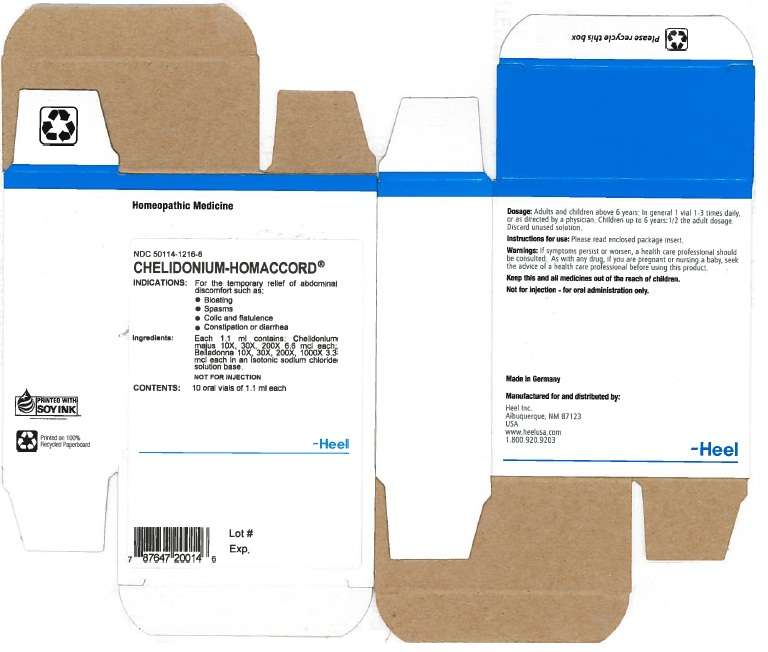
Chelidonium Homaccord
CHELIDONIUM MAJUS and ATROPA BELLADONNA SOLUTION
Product Information
|
|
Product Type
|
Human otc drug label |
Item Code (Source)
|
NDC:50114-1216 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
|
1.1 in 1 VIAL |
|
|
|
2 |
NDC:50114-1216-6 |
10 in 1 CARTON |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
|
|
1993-01-31 |
|
|
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!
