Visine Totality
Johnson & Johnson Healthcare Products, Division of McNEIL-PPC, Inc.
VISINE®Visine Totality Multi-Symptom Relief
FULL PRESCRIBING INFORMATION: CONTENTS*
- Visine Totality Uses
- Warnings
- Directions
- Visine Totality Other information
- Inactive ingredients
- Questions?
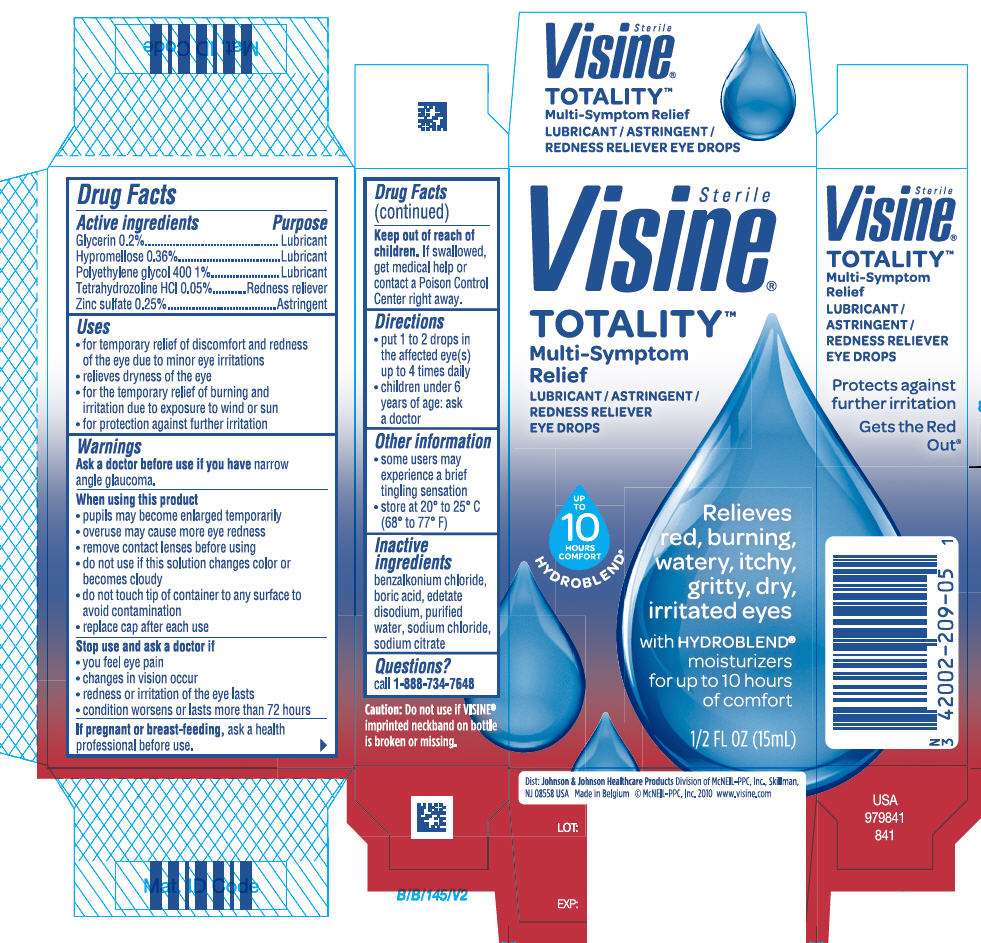
- PRINCIPAL DISPLAY PANEL - 15mL Bottle Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
Purpose
| Active ingredients | Purpose |
|---|---|
| Glycerin 0.2% | Lubricant |
| Hypromellose 0.36% | Lubricant |
| Polyethylene glycol 400 1% | Lubricant |
| Tetrahydrozoline HCl 0.05% | Redness reliever |
| Zinc sulfate 0.25% | Astringent |
Visine Totality Uses
- for temporary relief of discomfort and redness of the eye due to minor eye irritations
- relieves dryness of the eye
- for the temporary relief of burning and irritation due to exposure to wind or sun
- for protection against further irritation
Warnings
Ask a doctor before use if you have narrow angle glaucoma.
When using this product
- pupils may become enlarged temporarily
- overuse may cause more eye redness
- remove contact lenses before using
- do not use if this solution changes color or becomes cloudy
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Stop use and ask a doctor if
- you feel eye pain
- changes in vision occur
- redness or irritation of the eye lasts
- condition worsens or lasts more than 72 hours
If pregnant or breast feeding, ask a health professional before use.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- put 1 to 2 drops in the affected eye(s) up to 4 times daily
- children under 6 years of age: ask a doctor
Visine Totality Other information
- some users may experience a brief tingling sensation
- store at 20° to 25°C (68° to 77°F)
Inactive ingredients
benzalkonium chloride, boric acid, edetate disodium, purified water, sodium chloride, sodium citrate
Questions?
call 1-888-734-7648
Dist: Johnson & Johnson Healthcare Products Division of McNEIL-PPC, Inc., Skillman,
NJ 08558 USA
PRINCIPAL DISPLAY PANEL - 15mL Bottle Carton
Sterile
Visine•
TOTALITY™
Multi-Symptom
Relief
LUBRICANT / ASTRINGENT /
REDNESS RELIEVER
EYE DROPS
UP
TO
10
HOURS
COMFORT
HYDROBLEND•
Relieves
red, burning,
watery, itchy,
gritty, dry,
irritated eyes
with HYDROBLEND•
moisturizers
for up to 10 hours
of comfort
1/2FL OZ (15mL)
Visine TotalityGlycerin, Hypromelloses, Polyethylene Glycol 400, Tetrahydrozoline Hydrochloride, and Zinc Sulfate LIQUID
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||