Trihexyphenidyl Hydrochloride
FULL PRESCRIBING INFORMATION: CONTENTS*
- TRIHEXYPHENIDYL HYDROCHLORIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- WARNINGS
- PRECAUTIONS
- TRIHEXYPHENIDYL HYDROCHLORIDE ADVERSE REACTIONS
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- STORAGE AND HANDLING
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
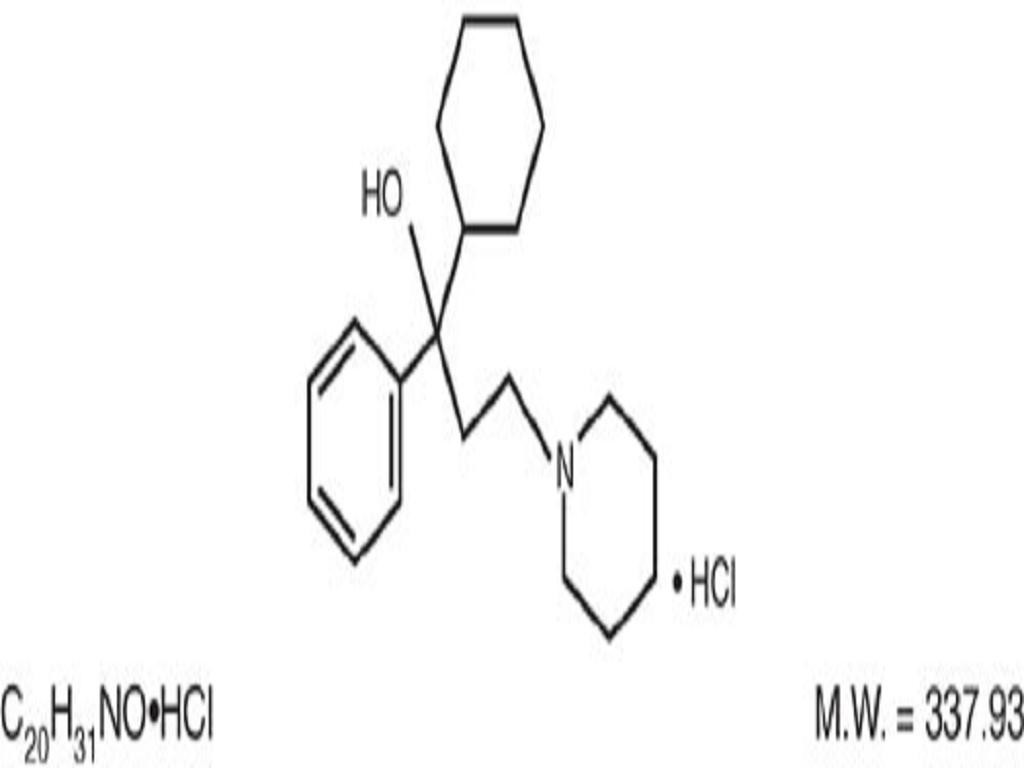
TRIHEXYPHENIDYL HYDROCHLORIDE DESCRIPTION
CLINICAL PHARMACOLOGY
INDICATIONS & USAGE
WARNINGS
PRECAUTIONS
TRIHEXYPHENIDYL HYDROCHLORIDE ADVERSE REACTIONS
DOSAGE & ADMINISTRATION
Trihexyphenidyl HCl in Idiopathic Parkinsonism
Trihexyphenidyl HCl in Drug-Induced Parkinsonism
Concomitant Use of Trihexyphenidyl HCl with Levodopa
Concomitant Use of Trihexyphenidyl HCl with Other Parasympathetic Inhibitors
HOW SUPPLIED
STORAGE AND HANDLING
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION


Trihexyphenidyl HydrochlorideTrihexyphenidyl Hydrochloride TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!