HYDROXYZINE HYDROCHLORIDE
HydrOXYzine Hydrochloride Tablets USP Revised: June 2009 Rx only 190944
FULL PRESCRIBING INFORMATION
Hydroxyzine hydrochloride is designated chemically as (±)-2-[2-[4-(p- Chloro-α-phenylbenzyl)-1-piperazinyl]ethoxy]ethanol dihydrochloride.
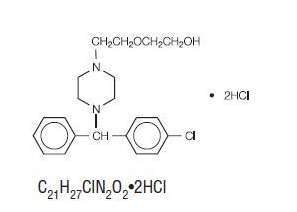
Hydroxyzine hydrochloride occurs as a white, odorless powder which is very soluble in water.
Hydroxyzine Hydrochloride Tablets USP, 10 mg, 25 mg and 50 mg contain the following inactive ingredients: anhydrous lactose, colloidal silicon dioxide, croscarmellose sodium, FD&C Yellow No. 6, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80 and titanium dioxide.
Hydroxyzine Hydrochloride Tablets USP, 25 mg and 50 mg also contain D&C Yellow No. 10 and FD&C Blue No. 2.
Hydroxyzine hydrochloride is unrelated chemically to the phenothiazines, reserpine, meprobamate, or the benzodiazepines. Hydroxyzine hydrochloride is not a cortical depressant, but its action may be due to a suppression of activity in certain key regions of the subcortical area of the central nervous system.
Primary skeletal muscle relaxation has been demonstrated experimentally. Bronchodilator activity, and antihistaminic and analgesic effects have been demonstrated experimentally and confirmed clinically. An antiemetic effect, both by the apomorphine test and the veriloid test, has been demonstrated.
Pharmacological and clinical studies indicate that hydroxyzine in therapeutic dosage does not increase gastric secretion or acidity and in most cases has mild antisecretory activity.
Hydroxyzine is rapidly absorbed from the gastrointestinal tract and hydroxyzine hydrochloride’s clinical effects are usually noted within 15 to 30 minutes after oral administration.
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested.
Useful in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses, and in histaminemediated pruritus.
As a sedative when used as premedication and following general anesthesia, hydroxyzine may potentiate meperidine and barbiturates, so their use in pre-anesthetic adjunctive therapy should be modified on an individual basis. Atropine and other belladonna alkaloids are not affected by the drug. Hydroxyzine is not known to interfere with the action of digitalis in any way and it may be used concurrently with this agent.
The effectiveness of hydroxyzine as an antianxiety agent for long term use, that is more than 4 months, has not been assessed by systematic clinical studies. The physician should reassess periodically the usefulness of the drug for the individual patient.
Hydroxyzine, when administered to the pregnant mouse, rat, and rabbit, induced fetal abnormalities in the rat and mouse at doses substantially above the human therapeutic range. Clinical data in human beings are inadequate to establish safety in early pregnancy. Until such data are available, hydroxyzine is contraindicated in early pregnancy.
Hydroxyzine is contraindicated for patients who have shown a previous hypersensitivity to it.
THE POTENTIATING ACTION OF HYDROXYZINE MUST BE CONSIDERED WHEN THE DRUG IS USED IN CONJUNCTION WITH CENTRAL NERVOUS SYSTEM DEPRESSANTS SUCH AS NARCOTICS, NONNARCOTIC ANALGESICS AND BARBITURATES. Therefore, when central nervous system depressants are administered concomitantly with hydroxyzine their dosage should be reduced.
Since drowsiness may occur with use of this drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery while taking hydroxyzine hydrochloride. Patients should be advised against the simultaneous use of other CNS depressant drugs, and cautioned that the effect of alcohol may be increased.
It is not known whether this drug is excreted in human milk. Since many drugs are so excreted, hydroxyzine should not be given to nursing mothers.
A determination has not been made whether controlled clinical studies of hydroxyzine included sufficient numbers of subjects aged 65 and over to define a difference in response from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
The extent of renal excretion of hydroxyzine has not been determined. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selections.
Sedating drugs may cause confusion and over sedation in the elderly; elderly patients generally should be started on low doses of hydroxyzine and observed closely.
Side effects reported with the administration of hydroxyzine hydrochloride are usually mild and transitory in nature.
Anticholinergic: Dry mouth.
Central Nervous System: Drowsiness is usually transitory and may disappear in a few days of continued therapy or upon reduction of the dose. Involuntary motor activity including rare instances of tremor and convulsions have been reported, usually with doses considerably higher than those recommended. Clinically significant respiratory depression has not been reported at recommended doses.
The most common manifestation of hydroxyzine hydrochloride overdosage is hypersedation. As in the management of overdosage with any drug, it should be borne in mind that multiple agents may have been taken.
If vomiting has not occurred spontaneously, it should be induced. Immediate gastric lavage is also recommended. General supportive care, including frequent monitoring of the vital signs and close observation of the patient, is indicated. Hypotension, though unlikely, may be controlled with intravenous fluids and norepinephrine or metaraminol. Do not use epinephrine as hydroxyzine hydrochloride counteracts its pressor action.
There is no specific antidote. It is doubtful that hemodialysis would be of any value in the treatment of overdosage with hydroxyzine. However, if other agents such as barbiturates have been ingested concomitantly, hemodialysis may be indicated. There is no practical method to quantitate hydroxyzine in body fluids or tissue after its ingestion or administration.
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested: in adults, 50-100 mg q.i.d.; children under 6 years, 50 mg daily in divided doses and over 6 years, 50-100 mg daily in divided doses.
For use in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses, and in histamine-mediated pruritus: in adults, 25 mg t.i.d. or q.i.d.; children under 6 years, 50 mg daily in divided doses and over 6 years, 50-100 mg daily in divided doses.
As a sedative when used as a premedication and following general anesthesia: 50-100 mg in adults, and 0.6 mg/kg of body weight in children.
When treatment is initiated by the intramuscular route of administration, subsequent doses may be administered orally.
As with all medications, the dosage should be adjusted according to the patient’s response to therapy.
Hydroxyzine Hydrochloride Tablets USP, 25 mg are 7/32”, unscored, round, green, film-coated tablets imprinted DAN and 5523 supplied in bottles of 30.
Dispense in a tight container with child-resistant closure.
Store at 20°-25°C (68°-77°F). [See USP controlled room temperature.]
Manufactured By:
Watson Pharma Private Ltd.
Verna, Salcette Goa 403 722 INDIA
Distributed By:
Watson Pharma, Inc.
Corona, CA 92880 USA
Revised: June 2009 190944
0609B
HYDROXYZINE HYDROCHLORIDEHYDROXYZINE HYDROCHLORIDE TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||