GUNA-SINUS NOSE SPRAY
DRUG FACTS
FULL PRESCRIBING INFORMATION: CONTENTS*
FULL PRESCRIBING INFORMATION
ACTIVE INGREDIENTS/PURPOSE
ALIUM CEPA 6X, 8X, 12X EXPECTORANT
ALTHEAS OFFICINALIS 2X EXPECTORANT
ARGENTUM NITRICUM 10X HEADACHE ANALGESIC
ASCORBIC ACID 2X ANTIOXIDANT
BLACK CURRANT, BARK 1X DETOXIFICATION
COMMON LUNGWORT 2X DECONGESTANT
CUPRUM METALLICUM 3X ANTIVIRAL IMMUNE SUPPORT
ECHINACEA ANGUSTIFOLIA 3X ANTIBACTERIAL IMMUNE SUPPORT
HEPAR SULFURIS CALCAREUM 6X, 8X, 12X ANTIOXIDANT
HYDRASTIS CANADENSIS 6X, 8X, 12X MUCOLYTIC
KALI BICHROMICUM 6X, 8X, 12X MUCOLYTIC
LACHESIS MUTUS 12X, 30X, 200X DECONGESTANT
MANGANUM METALLICUM 3X DECONGESTANT
MERCURIUS SULPHURATUS RUBER 6X, 8X, 12X ANTI-INFLAMMATORY
MUCOSA NASALIS 6X, 12X, 30X SUPPORTS IMMUNE RESPONSE
PLANTAGO MAJOR 2X DETOXIFICATION
PULSATILLA 6X, 8X, 12X DECONGESTANT
SILICEA 6X, 8X, 12X DECONGESTANT
SINUSITISINUM 12X, 30X DECONGESTANT
THYMUS GLAND 6X, 12X, 30X SUPPORTS IMMUNE RESPONSE
URINARY BLADDER 10X, 30X, 200X DETOXIFICATION
USES
For the temporary relief of symptoms due to acute and chronic Sinus Infection: Sinus Headaches, Post Nasal Drip, Nasal Congestion
WARNINGS
Stop use and ask doctor if symptoms of headache, nasal drip or nasal congestion worsen or persist more than 5 days
PREGNANCY
If pregnant or breast-feeding ask a doctor before use
WARNINGS
Keep this and all medicines out of reach of children
DIRECTIONS
Tilt head slightly forward, place tip of bottle into nose, compress bottle once for each spray, aiming towards the back of the nose
Adults and children 12 years and older 2 sprays to each nostril, 3-5 times per day
Children between 12 years and 6 years of 1 age 1 spray to each nostril, 3-5 times per day
Children under 6 years 1 spray to each nostril, 3 times per day
QUESTIONS
Questions?: info@gunainc.com
Tel. (484) 223-3500
PRINCIPAL DISPLAY PANEL
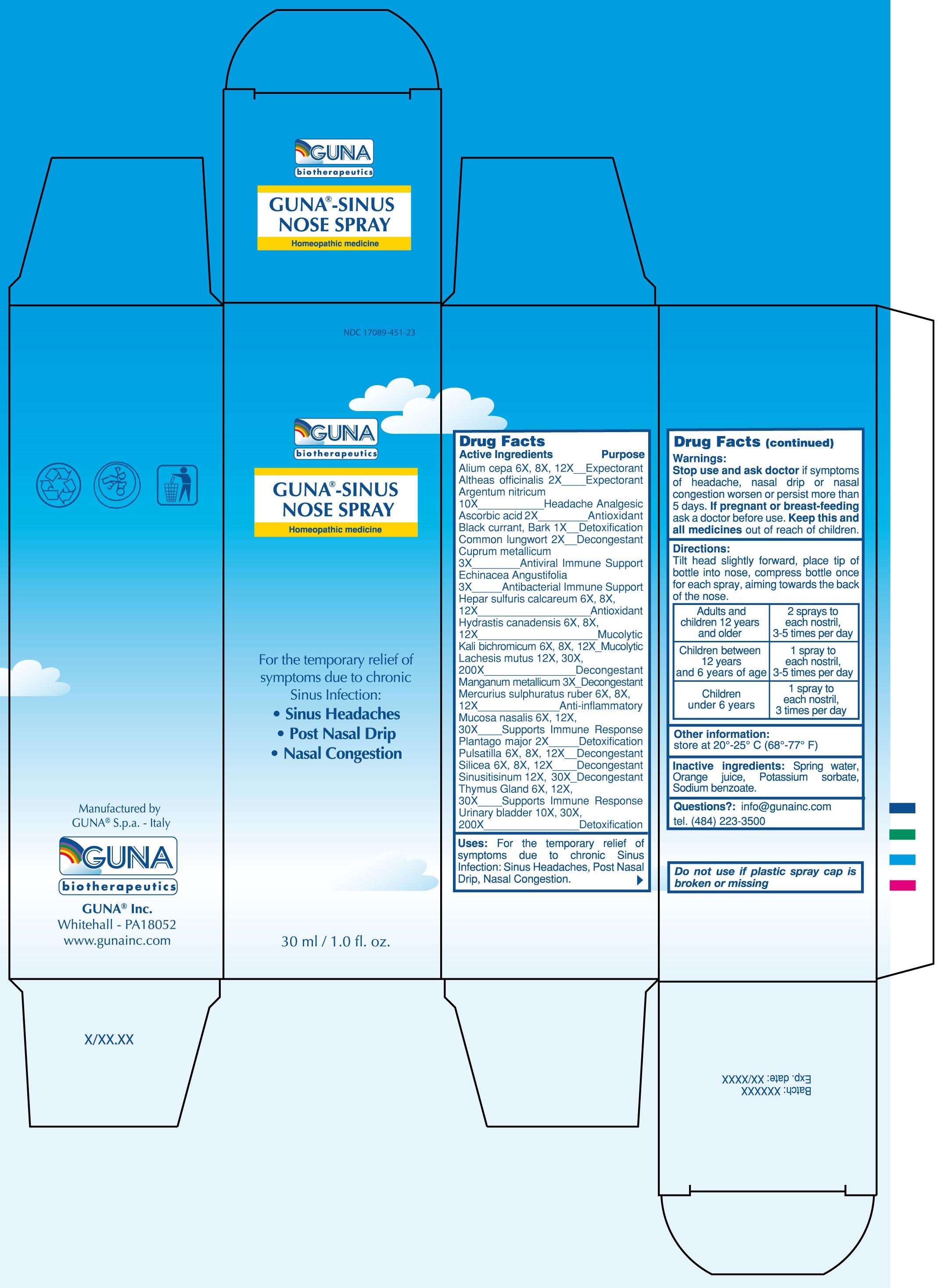
GUNA-SINUS NOSE SPRAYALTHAEA OFFICINALIS LEAF - ASCORBIC ACID - BLACK CURRANT - CALCIUM SULFIDE - COPPER - ECHINACEA ANGUSTIFOLIA - GOLDENSEAL - LACHESIS MUTA VENOM - LOBARIA PULMONARIA - MANGANESE - MERCURIC SULFIDE - ONION - PLANTAGO MAJOR - POTASSIUM CARBONATE - PULSATILLA VULGARIS - SILICON DIOXIDE - SILVER NITRATE - SINUSITISINUM - SUS SCROFA NASAL MUCOSA - SUS SCROFA THYMUS - SUS SCROFA URINARY BLADDER - SPRAY
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||