Doxepin Hydrochloride
FULL PRESCRIBING INFORMATION: CONTENTS*
- BOXED WARNING
- DOXEPIN HYDROCHLORIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- DOXEPIN HYDROCHLORIDE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- DRUG INTERACTIONS
- PEDIATRIC USE
- GERIATRIC USE
- DOXEPIN HYDROCHLORIDE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- STORAGE AND HANDLING
- SPL MEDGUIDE
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
BOXED WARNING
Suicidality and Antidepressant DrugsAntidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of doxepin or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyone age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Doxepin is not approved for use in pediatric patients. (See WARNINGS: Clinical Worsening and Suicide Risk,PRECAUTIONS: Information for PatientsandPRECAUTIONS: Pediatric Use.)
DOXEPIN HYDROCHLORIDE DESCRIPTION
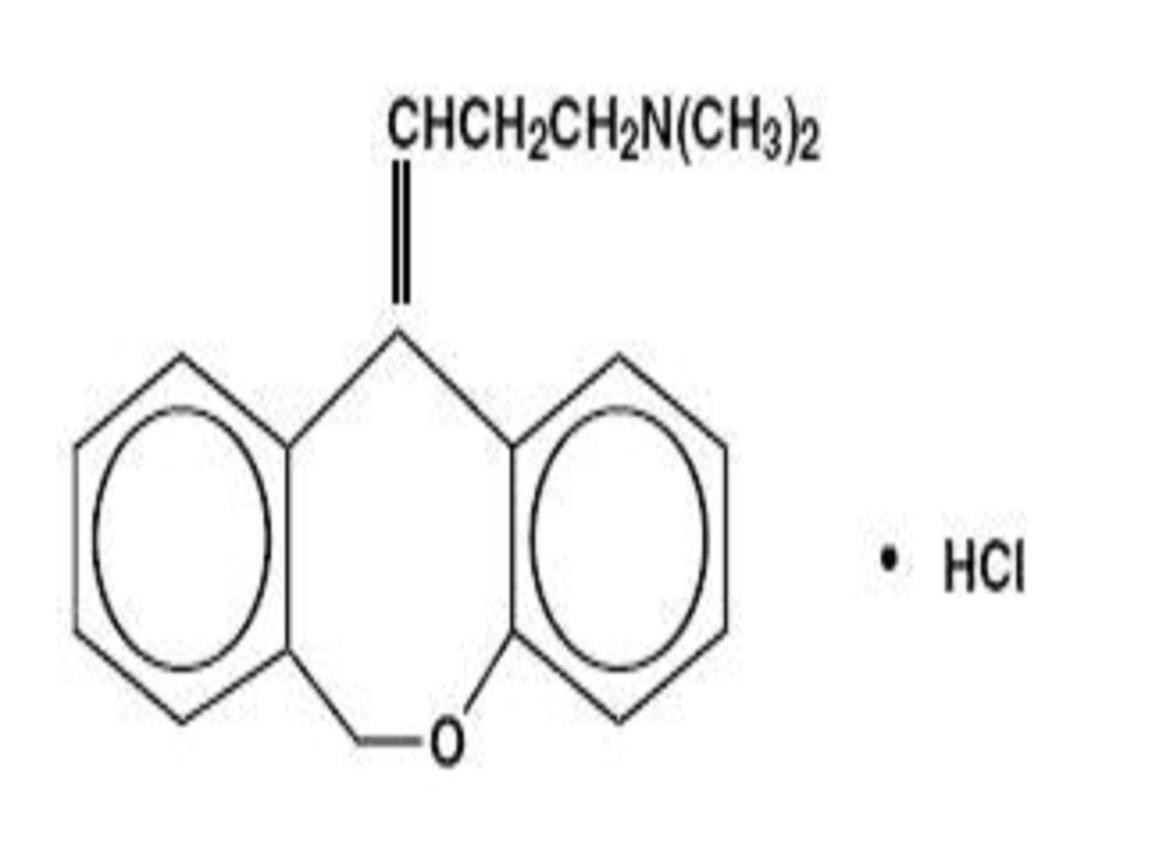
CLINICAL PHARMACOLOGY
INDICATIONS & USAGE
DOXEPIN HYDROCHLORIDE CONTRAINDICATIONS
WARNINGS
Clinical Worsening and Suicide Risk
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to healthcare providers. Such monitoring should include daily observation by families and caregivers.
Screening Patients for Bipolar Disorder
Geriatric Use
PRECAUTIONS: Geriatric Use
Pregnancy
Pediatric Use
PRECAUTIONS
Information for PatientsClinical Worsening and Suicide Risk
DRUG INTERACTIONS
Drugs Metabolized by P450 2D6MAO Inhibitors
Cimetidine
Alcohol
Tolazamide
Drowsiness
PRECAUTIONS: Geriatric Use
Suicide
Psychosis
PEDIATRIC USE
BOX WARNINGWARNINGS: Clinical Worsening and Suicide RiskGERIATRIC USE
WARNINGS
DOXEPIN HYDROCHLORIDE ADVERSE REACTIONS
NOTE:Anticholinergic Effects:
Central Nervous System Effects:
Cardiovascular:
Allergic:
Hematologic:
Gastrointestinal:
Endocrine:
Other:
Withdrawal Symptoms
OVERDOSAGE
Manifestations
ADVERSE REACTIONS
General Recommendations
General
Gastrointestinal Decontamination
Cardiovascular
CNS
Psychiatric Follow-up
Pediatric Management
DOSAGE & ADMINISTRATION
HOW SUPPLIED
STORAGE AND HANDLING
SPL MEDGUIDE
Antidepressant Medicines, Depression and Other Serious Mental Illnesses, and Suicidal Thoughts or ActionsTalk to your, or your family member's, healthcare provider about:
-
● all risks and benefits of treatment with antidepressant medicines
-
● all treatment choices for depression or other serious mental illness
-
● 1.Antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, and young adults within the first few months of treatment.
-
● 2.Depression and other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a particularly high risk of having suicidal thoughts or actions. These include people who have (or have a family history of) bipolar illness also called manic-depressive illness) or suicidal thoughts or actions.
-
● 3.How can I watch for and try to prevent suicidal thoughts and actions in myself or a family member?
-
● Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
-
● Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
-
● Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
What else do I need to know about antidepressant medicines?
-
● Never stop an antidepressant medicine without first talking to a healthcare provider.Stopping an antidepressant medicine suddenly can cause other symptoms.
-
● Antidepressants are medicines used to treat depression and other illnesses.It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients and their families or other caregivers should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
-
● Antidepressant medicines have other side effects.Talk to the healthcare provider about the side effects of the medicine prescribed for you or your family member.
-
● Antidepressant medicines can interact with other medicines. Know all of the medicines that you or your family member takes. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
-
● Not all antidepressant medicines prescribed for children are FDA approved for use in children.Talk to your child's healthcare provider for more information.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

Doxepin HydrochlorideDoxepin Hydrochloride CAPSULE
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!