Doxazosin Mesylate
FULL PRESCRIBING INFORMATION: CONTENTS*
- DOXAZOSIN MESYLATE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACODYNAMICS
- PHARMACOKINETICS
- INDICATIONS & USAGE
- DOXAZOSIN MESYLATE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- DRUG & OR LABORATORY TEST INTERACTIONS
- DRUG INTERACTIONS
- ANIMAL PHARMACOLOGY & OR TOXICOLOGY
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- DOXAZOSIN MESYLATE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- STORAGE AND HANDLING
- SPL PATIENT PACKAGE INSERT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
DOXAZOSIN MESYLATE DESCRIPTION

CLINICAL PHARMACOLOGY
PHARMACODYNAMICS
Benign Prostatic Hyperplasia (BPH)TABLE 1 SUMMARY OF EFFECTIVENESS DATA IN PLACEBO-CONTROLLED TRIALS
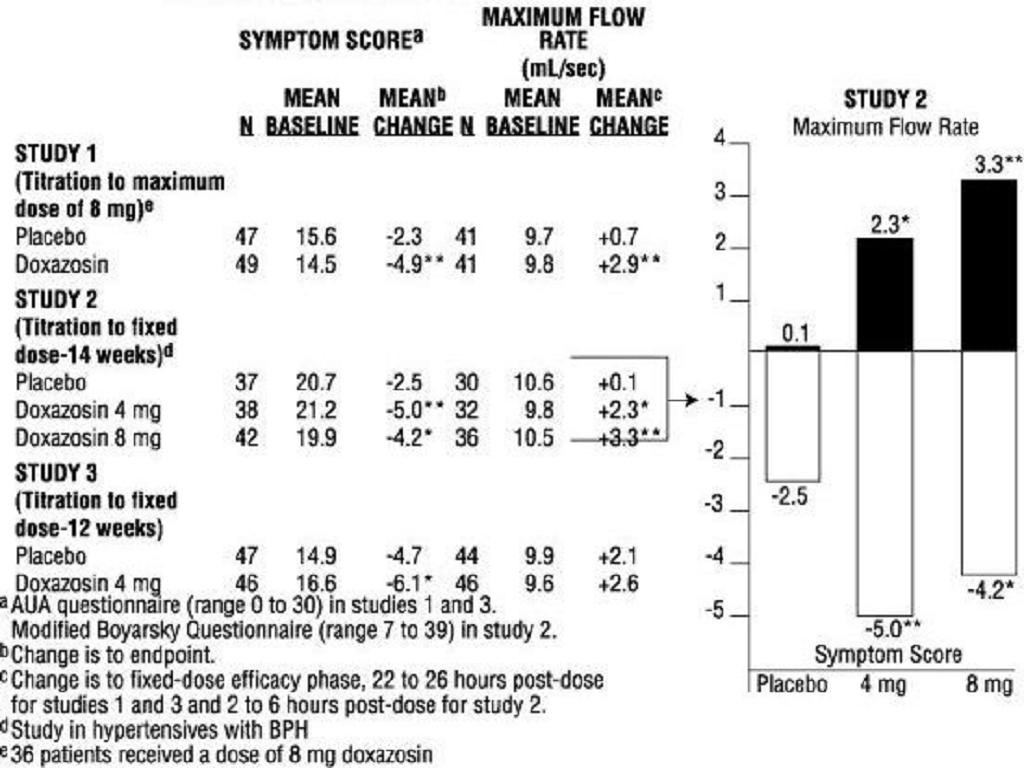
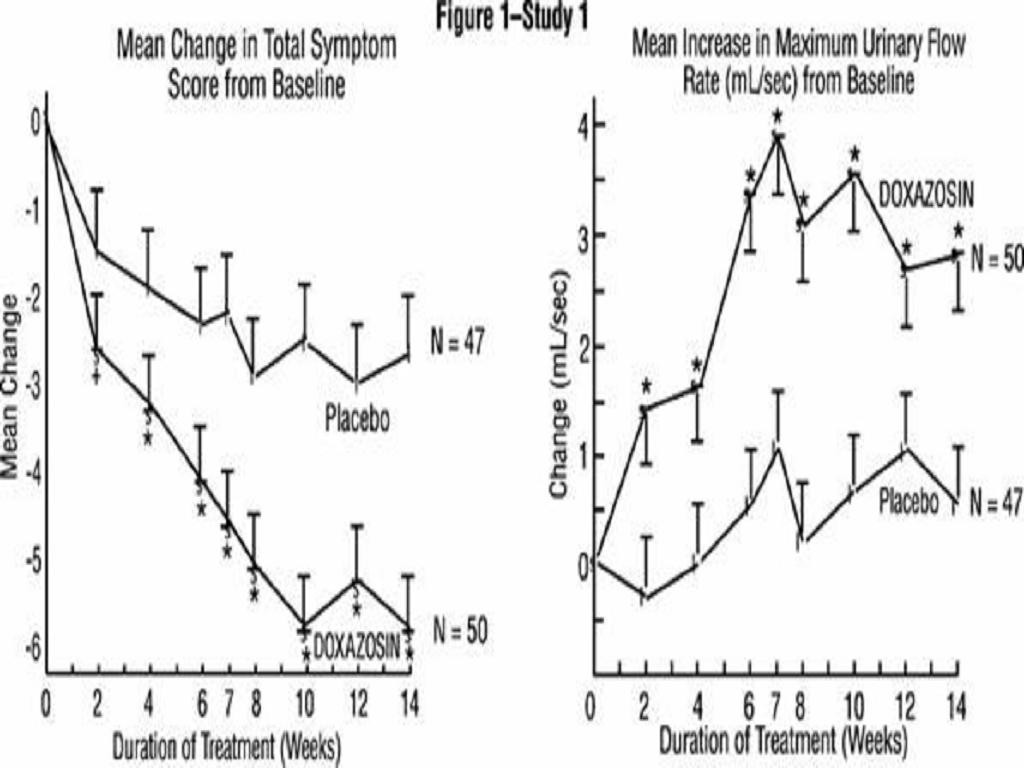
*****
Hypertension
PHARMACOKINETICS
PRECAUTIONS: Drug Interactions
INDICATIONS & USAGE
Benign Prostatic Hyperplasia (BPH)Hypertension
DOXAZOSIN MESYLATE CONTRAINDICATIONS
WARNINGS
Syncope and "First-dose" EffectDOSAGE AND ADMINISTRATION
Priapism
PRECAUTIONS: Information for Patients
PRECAUTIONS
GeneralProstate Cancer
Cataract Surgery
Orthostatic Hypotension
Hypertension
Benign Prostatic Hyperplasia
INFORMATION FOR PATIENTS
Patient LeafletDRUG & OR LABORATORY TEST INTERACTIONS
Impaired Liver Function
CLINICAL PHARMACOLOGY: Pharmacokinetics
Leukopenia/Neutropenia
DRUG INTERACTIONS
DOSAGE AND ADMINISTRATION
ANIMAL PHARMACOLOGY & OR TOXICOLOGY
Cardiac Toxicity in AnimalsCARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
Teratogenic Effects. Pregnancy Category CNonteratogenic Effects
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
For Hypertension
DOXAZOSIN MESYLATE ADVERSE REACTIONS
Benign Prostatic Hyperplasia (BPH)******
Hypertension
PRECAUTIONS: Leukopenia/Neutropenia
PRECAUTIONS: General: Cataract Surgery
OVERDOSAGE
DOSAGE & ADMINISTRATION
Benign Prostatic Hyperplasia 1 mg to 8 mg Once Daily
Hypertension 1 mg to 16 mg Once Daily
HOW SUPPLIED
STORAGE AND HANDLING
SPL PATIENT PACKAGE INSERT
PATIENT INFORMATION ABOUT DOXAZOSIN TABLETS, USP(dox az'oh sin)
FOR BENIGN PROSTATIC HYPERPLASIA (BPH)
-
● before you start taking doxazosin tablets.
-
● each time you get a new prescription.
-
● a weak or start-and-stop stream when urinating
-
● a feeling that the bladder is not completely emptied after urination
-
● a delay or difficulty in the beginning of urination
-
● a need to urinate often during the day and especially at night
-
● a feeling that you must urinate immediately.
-
● If you are not bothered by your symptoms, you and your doctor may decide on a program of "watchful waiting". It is not an active treatment like taking medication or surgery but involves having regular checkups to see if your condition is getting worse or causing problems.
-
● Treatment with doxazosin tablets or other similar drugs. Doxazosin tablets is the medication your doctor has prescribed for you. See "What Doxazosin Tablets Do", below.
-
● Treatment with the medication class of 5-alpha reductase inhibitors (e.g., finasteride). It can cause the prostate to shrink. It may take 6 months or more for the full benefit of finasteride to be seen.
-
● Various surgical procedures. Your doctor can describe these procedures to you. The best procedure for you depends on your BPH symptoms and medical condition.
-
●
-
● If doxazosin tablets are helping you, you should notice an affect within 1 to 2 weeks after you start your medication. Doxazosin tablets have been studied in over 900 patients for up to 2 years and the drug has been shown to continue to work during long-term treatment. Even though you take doxazosin tablets and it may help you, doxazosin tablets may not prevent the need for surgery in the future.
-
● Doxazosin tablets do not affect PSA levels. PSA is the abbreviation for Prostate Specific Antigen. Your doctor may have done a blood test called PSA. You may want to ask your doctor more about this if you have had a PSA test done.
-
● You should see an improvement of your symptoms within 1 to 2 weeks. In addition to your other regular checkups you will need to continue seeing your doctor regularly to check your progress regarding your BPH and to monitor your blood pressure.
-
● Doxazosin Mesylate is not a treatment for prostate cancer. Your doctor has prescribed doxazosin tablets for your BPH and not for prostate cancer; however, a man can have BPH and prostate cancer at the same time. Doctors usually recommend that men be checked for prostate cancer once a year when they turn 50 (or 40 if a family member has had prostate cancer). A higher incidence of prostate cancer has been noted in men of African-American descent. These checks should continue even if you are taking doxazosin tablets.
-
●
-
● Other side effects you could have while taking doxazosin tablets, in addition to lowering of the blood pressure, include dizziness, fatigue (tiredness), swelling of the feet and shortness of breath. Most side effects are mild. However, you should discuss any unexpected effects you notice with your doctor.
-
● WARNING: Extremely rarely, doxazosin tablets and similar medications have caused painful erection of the penis, sustained for hours and unrelieved by sexual intercourse or masturbation. This condition is serious, and if untreated it can be followed by permanent inability to have an erection. If you have a prolonged abnormal erection, call your doctor or go to an emergency room as soon as possible.
-
● Tell your surgeon if you take or have taken doxazosin tablets if you plan to have surgery for cataracts (clouding of the eye). During cataract surgery, a condition called Intraoperative Floppy Iris Syndrome (IFIS) can happen if you take or have taken doxazosin tablets.
-
● If you use doxazosin tablets with an oral erectile dysfunction medicine (phosphodiesterase-5 (PDE-5) inhibitor), it can cause a sudden drop in your blood pressure and you can become dizzy or faint. Talk with your healthcare provider before using PDE-5 inhibitors.
-
● Keep doxazosin tablets and all medicines out of the reach of children.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

Doxazosin MesylateDoxazosin Mesylate TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!