Dopamine Hydrochloride
General Injectables & Vaccines, Inc
Dopamine HCL 40 mg/mL Injection, USP 10 mL Single Dose Vial
FULL PRESCRIBING INFORMATION: CONTENTS*
- Description
- Clinical Pharmacology
- Dopamine Hydrochloride Indications and Usage
- Contraindications
- Warnings
- Precautions
- Side Effects
- Overdosage
- Dosage and Administration
- How Supplied
- Sample Outer Label
FULL PRESCRIBING INFORMATION
Description
Dopamine, a sympathomimetic amine vasopressor, is the naturally occurring immediate precursor of norepinephrine. Dopamine hydrochloride is a white to off-white crystalline powder, which may have a slight odor of hydrochloric acid. It is freely soluble in water and soluble in alcohol. Dopamine HCl is sensitive to alkalies, iron salts, and oxidizing agents. Chemically it is designated as 4-
(2-aminoethyl) pyrocatechol hydrochloride, and its molecular formula is C8H11NO2 • HCl. The structural formula is:
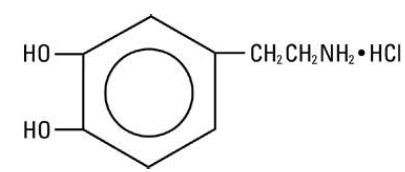
and the molecular weight is 189.64. Dopamine hydrochloride injection is a clear, practically colorless, sterile, pyrogen-free, aqueous solution of dopamine HCl for intravenous infusion after dilution. Each milliliter of the 40 mg/mL preparation contains 40 mg of dopamine hydrochloride (equivalent to 32.31 mg of dopamine base). Each milliliter of the 80 mg/mL preparation contains 80 mg of dopamine hydrochloride (equivalent to 64.62 mg of dopamine base). Each milliliter of both preparations contains the following: Sodium metabisulfite 9 mg added as an antioxidant; citric acid, anhydrous 10 mg; and sodium citrate, dihydrate 5 mg added as a buffer. May contain additional citric acid and/ or sodium citrate for pH adjustment. pH is 3.3 (2.5 to 5.0). Dopamine must be diluted in an appropriate sterile parenteral solution before intravenous administration. (See DOSAGE AND ADMINISTRATION)
Clinical Pharmacology
Dopamine is a natural catecholamine formed by the decarboxylation of 3,4-dihydroxyphenylalanine (DOPA). It is a precursor to norepinephrine in noradrenergic nerves and is also a neurotransmitter in certain areas of the central nervous system, especially in the nigrostriatal tract, and in a few peripheral sympathetic nerves. Dopamine produces positive chronotropic and inotropic effects on the myocardium, resulting in increased heart rate and cardiac contractility. This is accomplished directly by exerting an agonist action on beta-adrenoceptors and indirectly by causing release of norepinephrine from storage sites in sympathetic nerve endings. Dopamine’s onset of action occurs within five minutes of intravenous administration, and with dopamine’s plasma half-life of about two minutes, the duration of action is less than ten minutes. However, if monoamine oxidase (MAO) inhibitors are present, the duration may increase to one hour. The drug is widely distributed in the body but does not cross the blood-brain barrier to a significant extent. Dopamine is metabolized in the liver, kidney, and plasma by MAO and catechol-O-methyltransferase to the inactive compounds homovanillic acid (HVA) and 3,4-dihydroxyphenylacetic acid. About 25% of the dose is taken up into specialized neurosecretory vesicles (the adrenergic nerve terminals), where it is hydroxylated to form norepinephrine. It has been reported that about 80% of the drug is excreted in the urine within 24 hours, primarily as HVA and its sulfate and glucuronide conjugates and as 3,4-dihydroxyphenylacetic acid. A very small portion is excreted unchanged. The predominant effects of dopamine are dose-related, although it should be noted that actual response of an individual patient will largely depend on the clinical status of the patient at the time the drug is administered. At low rates of infusion (0.5 - 2 mcg/kg/min) dopamine causes vasodilation that is presumed to be due to a specific agonist action on dopamine receptors (distinct from alpha- and beta-adrenoceptors) in the renal, mesenteric, coronary, and intracerebral vascular beds. At these dopamine receptors, haloperidol is an antagonist. The vasodilation in these vascular beds is accompanied by increased glomerular filtrationrate, renal blood flow, sodium excretion, and urine flow. Hypotension sometimes occurs. An increase in urinary output produced by dopamine is usually not associated with a decrease in osmolality of the urine. At intermediate rates of infusion (2 - 10 mcg/kg/min) dopamine acts to stimulate the beta1-adrenoceptors, resulting in improved myocardial contractility, increased SA rate and enhanced impulse conduction in the heart. There is little, if any, stimulation of the beta2-adrenoceptors (peripheral vasodilation). Dopamine causes less increase in myocardial oxygen consumption than isoproterenol, and its use is not usually associated with a tachyarrhythmia. Clinical studies indicate that it usually increases systolic and pulse pressure with either no effect or a slight increase in diastolic pressure. Blood flow to the peripheral vascular beds may decrease while mesenteric flow increases due to increased cardiac output. Total peripheral resistance (alpha effects) at low and intermediate doses is usually unchanged. At higher rates of infusion (10 - 20 mcg/kg/min) there is some effect on alpha-adrenoceptors, with consequent vasoconstrictor effects and a rise in blood pressure. The vasoconstrictor effects are first seen in the skeletal muscle vascular beds, but with increasing doses, they are also evident in the renal and mesenteric vessels. At very high rates of infusion (above 20 mcg/kg/min), stimulation of alphaadrenoceptors predominates and vasoconstriction may compromise the circulation of the limbs and override the dopaminergic effects of dopamine, reversing renal dilation and naturesis.
Dopamine Hydrochloride Indications and Usage
Dopamine HCl is indicated for the correction of hemodynamic imbalances present in the shock syndrome due to myocardial infarction, trauma, endotoxic septicemia, open-heart surgery, renal failure, and chronic cardiac decompensation as in congestive failure.
Patients most likely to respond adequately to dopamine HCl are those in whom physiological parameters, such as urine flow, myocardial function, and blood pressure, have not undergone profound deterioration. Multiclinic trials indicate that the shorter the time interval between onset of signs and symptoms and initiation of therapy with blood volume correction and dopamine HCl, the better the prognosis. Where appropriate, blood volume restoration with a suitable plasma expander or whole blood should be accomplished prior to administration of dopamine HCl.
Poor Perfusion of Vital Organs- Urine flow appears to be one of the better diagnostic signs by which adequacy of vital organ perfusion can be monitored. Nevertheless, the physician should also observe the patient for signs of reversal of confusion or reversal of comatose condition. Loss of pallor, increase in toe temperature, and/or adequacy of nail bed capillary filling may also be used as indices of adequate dosage. Clinical studies have shown that when dopamine HCl is administered before urine flow has diminished to levels of approximately 0.3 mL/minute, prognosis is more favorable. Nevertheless, in a number of oliguric or anuric patients, administration of dopamine HCl has resulted in an increase in urine flow, which in some cases reached normal levels. Dopamine HCl may also increase urine flow in patients whose output is within normal limits and thus may be of value in reducing the degree of preexisting fluid accumulation. It should be noted that at doses above those optimal for the individual patient, urine flow may decrease, necessitating reduction of dosage.
Low Cardiac Output - Increased cardiac output is related to dopamine’s direct inotropic effect on the myocardium. Increased cardiac output at low or moderate doses appears to be related to a favorable prognosis. Increase in cardiac output has been associated with either static or decreased systemic vascular resistance (SVR). Static or decreased SVR associated with low or moderate movements in cardiac output is believed to be a reflection of differential effects on specific vascular beds with increased resistance in peripheral beds (e.g., femoral) and concomitant decreases in mesenteric and renal vascular beds. Redistribution of blood flow parallels these changes so that an increase in cardiac output is accompanied by an increase in mesenteric and renal blood flow. In many instances the renal fraction of the total cardiac output has been found to increase. Increase in cardiac output produced by dopamine is not associated with substantial decreases in systemic vascular resistance as may occur with isoproterenol.
Hypotension - Hypotension due to inadequate cardiac output can be managed by administration of low to moderate doses of dopamine HCl which have little effect on SVR. At high therapeutic doses, dopamine’s alpha-adrenergic activity becomes more prominent and thus may correct hypotension due to diminished SVR. As in the case of other circulatory decompensation states,
prognosis is better in patients whose blood pressure and urine flow have not undergone profound deterioration. Therefore, it is suggested that the physician administer dopamine HCl as soon as a definite trend toward decreased systolic and diastolic pressure becomes evident.
Contraindications
Dopamine HCl should not be used in patients with pheochromocytoma.
Dopamine HCl should not be administered to patients with uncorrected tachyarrhythmias or ventricular fibrillation.
Warnings
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Do NOT add dopamine HCl to any alkaline diluent solution since the drug is inactivated in alkaline solution. Patients who have been receiving MAO inhibitors prior to the administration of dopamine HCl will require substantially reduced dosage. See Drug Interactions below.
Precautions
General
1. Monitoring - Careful monitoring of the following indices is necessary during dopamine HCl infusion, as with any adrenergic agent: blood pressure, urine flow, and, when possible, cardiac output and pulmonary wedge pressure.
2. Hypovolemia - Prior to treatment with dopamine HCl, hypovolemia should be fully corrected, if possible, with either whole blood or plasma as indicated. Monitoring of central venous pressure or left ventricular filling pressure may be helpful in detecting and treating hypovolemia.
3. Hypoxia
, Hypercapnia, Acidosis - These conditions, which may also reduce the effectiveness and/or increase the incidence of adverse effects of dopamine, must be identified and corrected prior to, or concurrently with, administration of dopamine HCl.
4. Decreased Pulse Pressure - If a disproportionate increase in diastolic blood pressure and a marked decrease in pulse pressure are observed in patients receiving dopamine HCl, the rate of infusion should be decreased and the patient observed carefully for further evidence of predominant vasoconstrictor activity, unless such effect is desired.
5. Ventricular Arrhythmias - If an increased number of ectopic beats are observed, the dose should be reduced if possible.
6. Hypotension - At lower infusion rates, if hypotension occurs, the infusion rate should be rapidly increased until adequate blood pressure is obtained. If hypotension persists, dopamine HCl should be discontinued and a more potent vasoconstrictor agent such as norepinephrine should be administered.
7. Extravasation - Dopamine HCl should be infused into a large vein whenever possible to prevent the possibility of extravasation into tissue adjacent to the infusion site. Extravasation may cause necrosis and sloughing of surrounding tissue. Large veins of the antecubital fossa are preferred to veins in the dorsum of the hand or ankle. Less suitable infusion sites should be used only if the patient’s condition requires immediate attention. The physician should switch to more suitable sites as rapidly as possible. The
infusion site should be continuously monitored for free flow.
8. Occlusive Vascular Disease - Patients with a history of occlusive vascular disease (for example, atherosclerosis, arterial embolism, Raynaud’s disease, cold injury, diabetic endarteritis, and Buerger’s disease) should be closely monitored for any changes in color or temperature of the skin in the extremities. If a change in skin color or temperature occurs and is thought to be the result of compromised circulation in the extremities, the benefits of continued dopamine HCl infusion should be weighed against the risk of possible necrosis. This condition may be reversed by either decreasing the rate or discontinuing the infusion.
IMPORTANTAntidote for Peripheral Ischemia: To prevent sloughing and necrosis in ischemic areas, the area should be infiltrated as soon as possible with 10 to 15 mL of saline solution containing from 5 to 10 mg of phentolamine mesylate, an adrenergic blocking agent. A syringe with a fine hypodermic needle should be used, and the solution liberally infiltrated throughout the ischemic area. Sympathetic blockade with phentolamine causes immediate and conspicuous local hyperemic changes if the area is infiltrated within 12 hours. Therefore, phentolamine should be given as soon as possible after the extravasation is noted.
Weaning
Drug Interactions
Pregnancy
Teratogenic Effects: Pregnancy Category C
Labor and Delivery
Nursing Mothers
Pediatric Use
Side Effects
The following adverse reactions have been observed, but there are not enough data to support an estimate of their frequency.
Cardiovascular System:
ventricular arrhythmia (at very high doses)
ectopic beats
tachycardia
anginal pain
palpitation
cardiac conduction abnormalities
widened QRS complex
bradycardia
hypotension
hypertension
vasoconstriction
Respiratory System:
dyspnea
Gastrointestinal System:
nausea
vomiting
Metabolic/Nutritional System:
azotemia
Central Nervous System:
headache
anxiety
Dermatological System:
piloerection
Other:
Gangrene of the extremities has occurred when high doses were administered for prolonged periods or in patients with occlusive vascular disease receiving low doses of dopamine HCl.
Overdosage
In the case of accidental overdosage, as evidenced by excessive elevation of blood pressure, reduce rate of administration or temporarily discontinue dopamine HCl until patient’s condition stabilizes. Since dopamine’s duration of action is quite short, no additional remedial measures are usually necessary. If these measures fail to stabilize the patient’s condition, use of the short-acting alpha-adrenergic blocking agent phentolamine should be considered.
Dosage and Administration
WARNING: This is a potent drug; it must be diluted before administration to the patient.
Dopamine Hydrochloride Injection, USP is administered (only after dilution) by intravenous infusion.
Suggested Dilution - For the 40 mg/mL preparation, transfer by aseptic technique the contents containing either 5 mL, 200 mg or 10 mL, 400 mg of Dopamine Hydrochloride to either a 250 mL or 500 mL bottle of one of the sterile I.V. solutions listed below. For the 80 mg/mL preparation, transfer by aseptic technique the contents containing 10 mL, 800 mg of Dopamine Hydrochloride to a 250 mL, 500 mL or 1000 mL bottle of one of the following sterile I.V. solutions:
0.9% Sodium Chloride Injection, USP
5% Dextrose Injection, USP
5% Dextrose and 0.9% Sodium Chloride Injection, USP
5% Dextrose and 0.45% Sodium Chloride Injection, USP
5% Dextrose and Lactated Ringer’s Injection
Sodium Lactate Injection, USP 1/6 Molar
Lactated Ringer’s Injection, USP
The resultant dilutions are summarized in the following chart:
|
Concentration of Dopamine Hydrochloride |
40 mg/mL |
|
80 mg/mL |
| Volume of Dopamine Hydrochloride Injection, USP |
5 mL |
10 mL |
10 mL |
| 250 mL Bottle of I.V. Solution |
800 mcg/mL |
1600 mcg/mL |
3200 mcg/mL |
| 500 mL Bottle of I.V. Solution |
400 mcg/mL |
800 mcg/mL |
1600 mcg/mL |
| 1000 mL Bottle of I.V. Solution |
200 mcg/mL |
400 mcg/mL |
800 mcg/mL |
Rate of Administration
Suggested Regimen:
How Supplied
Dopamine Hydrochloride Injection, USP is supplied as follows (40 mg/mL and 80 mg/mL):
| 5 mL Fliptop Vial (200 mg) |
List No. 5820 |
| 5 mL LifeShield Fliptop Vial |
List No. 5820 |
| 10 mL Flipt Vial (400 mg) |
List No. 9104 |
| 10 mL LifeShield Fliptop Vial* |
List No. 9104 |
| 10 mL FlipTop Vial (800 mg) |
List No. 4265 |
Sample Outer Label

Dopamine HydrochlorideDopamine Hydrochloride INJECTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||