Donepezil Hydrochloride
Macleods Pharmaceuticals Limited
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Donepezil Hydrochloride Orally Disintegrating Tablets USP safely and effectively. See full prescribing information for Donepezil hydrochloride ODT USP. Donepezil Hydrochloride Orally Disintegrating Tablets USPInitial U.S. Approval: 1996 DOSAGE AND ADMINISTRATION Mild to Moderate Alzheimer's Disease – 5mg or 10mg administered once daily (2.1). Severe Alzheimer's Disease – 10 mg administered once daily (2.2). 2.3CONTRAINDICATIONS Patients with known hypersensitivity to donepezil hydrochloride or piperidine derivatives (4). WARNINGS AND PRECAUTIONS Cholinesterase inhibitors are likely to exaggerate succinylchloline- type muscle relaxation during anesthesia (5.1) Cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes manifesting as bradycardia or heart block (5.2). Donepezil hydrochloride ODT can cause vomiting. Patients should be observed closely at initiation of treatment and after dose increases (5.3). Patients should be monitored closely for symptoms of active or occult gastrointestinal (GI)bleeding, especially those at increased risk for developing ulcers (5.4). Cholinomimetics may cause bladder overflow obstructions (5.6). Cholinomimetics are believed to have some potential to cause generalized convulsions (5.7). Cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease (5.8). Side Effects6.1To report SUSPECTED ADVERSE REACTIONS, contact Macleods Pharma USA, Inc. at 1-888-943-3210 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatchDRUG INTERACTIONS Cholinesterase inhibitors have the potential to interfere with the activity of anticholinergic medications (7.2). A synergistic effect may be expected with concomitant administration of succinylcholine, similar neuromuscular blocking agents, or cholinergic agonists (7.3). USE IN SPECIFIC POPULATIONS Based on animal data, donepezil hydrochloride ODT may cause fetal harm (8.1).
FULL PRESCRIBING INFORMATION: CONTENTS*
- 1 INDICATIONS & USAGE
- 2 DOSAGE & ADMINISTRATION
- 3 DOSAGE FORMS & STRENGTHS
- 4 DONEPEZIL HYDROCHLORIDE CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
- 6 DONEPEZIL HYDROCHLORIDE ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
- 8 USE IN SPECIFIC POPULATIONS
- 10 OVERDOSAGE
- 11 DONEPEZIL HYDROCHLORIDE DESCRIPTION
- 12 CLINICAL PHARMACOLOGY
- 13 NONCLINICAL TOXICOLOGY
- 14 CLINICAL STUDIES
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
FULL PRESCRIBING INFORMATION
1 INDICATIONS & USAGE
Donepezil hydrochloride USP is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer’s disease.
2 DOSAGE & ADMINISTRATION
Donepezil hydrochloride ODT should be taken in the evening just before retiring.
2.1 Mild to Moderate Alzheimer's Disease
The dosages of donepezil hydrochloride shown to be effective in controlled clinical trials are 5 mg and 10 mg administered once per day.
2.2Moderate to Severe Alzheimer's Disease
2.3 Titration
The recommended starting dose of donepezil hydrochloride is 5mg once daily. Evidence from the controlled trials in mild to moderate Alzheimer's disease indicates that the 10 mg dose, with a one week titration, is likely to be associated with a higher incidence of cholinergic adverse events compared to the 5 mg dose. In open-label trials using a 6 week titration, the type and frequency of these same adverse events were similar between the 5 mg and 10 mg dose groups. Therefore, because donepezil hydrochloride steady state is achieved about 15 days after it is started and because the incidence of untoward effects may be influenced by the rate of dose escalation, a dose of 10 mg should not be administered until patients have been on a daily dose of 5 mg for 4 to 6 weeks.
3 DOSAGE FORMS & STRENGTHS
Donepezil hydrochloride ODT is supplied as round tablets containing either 5 mg or 10 mg of donepezil hydrochloride.
The 5 mg orally disintegrating tablets are yellow coloured, circular, flat face beveled edge uncoated tablets debossed with "CL 31" on one side and plain on the other side.
The 10 mg orally disintegrating tablets are yellow coloured, circular, flat face beveled edge uncoated tablets debossed with "CL 32" on one side and plain on the other side.
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Anesthesia
5.2 Cardiovascular Conditions
5.3 Nausea and Vomiting
Donepezil hydrochloride, as a predictable consequence of its pharmacological properties, has been shown to produce diarrhea, nausea and vomiting. These effects, when they occur, appear more frequently with the 10mg/day dose than with the 5mg/day dose.
5.4 Peptic Ulcer disease and GI bleeding
Through their primary action, cholinesterase inhibitors may be expected to increase gastric acid secretion due to increased cholinergic activity. Therefore, patients should be monitored closely for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e.g., those with a history of ulcer disease or those receiving concurrent nonsteroidal anti-inflammatory drugs (NSAIDS).
5.6 Genitourinary Conditions
5.7 Neurological Conditions: Seizures
5.8 Pulmonary Conditions
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Donepezil hydrochloride orally disintegrating tablets 5 mg / day and 10 mg / day
Mild To Moderate Alzheimer's Disease
Adverse Events Leading to Discontinuation
|
Table 1. Most Frequent Adverse Events Leading to Discontinuation from Controlled Clinical Trials by Dose Group |
|||
|
Dose Group |
Placebo |
5 mg/day Donepezil hydrochloride |
10 mg/day Donepezil hydrochloride |
|
Patients Randomized
|
355 |
350 |
315 |
|
Event/%Discontinuing
|
|
|
|
|
Nausea |
1% |
1% |
3% |
|
Diarrhea
|
0% |
< 1% |
3% |
|
Vomiting |
<1% |
< 1% |
2% |
Most Frequent Adverse Clinical Events Seen in Association with the Use of Donepezil hydrochloride
|
Table 2. Comparison of rates of adverse events in mild to moderate patients titrated to 10 mg/day over 1 and 6 weeks |
||||
|
|
No titration |
One week titration |
Six week titration |
|
|
Adverse Event
|
Placebo (n=315) |
5 mg/day (n=311) |
10 mg/day (n=315) |
10 mg/day (n=269) |
| Nausea |
6% |
5% |
19% |
6% |
| Diarrhea |
5% |
8% |
15% |
9% |
| Insomnia |
6% |
6% |
14% |
6% |
| Fatigue |
3% |
4% |
8% |
3% |
| Vomiting |
3% |
3% |
8% |
5% |
| Muscle cramps |
2% |
6% |
8% |
3% |
| Anorexia |
2% |
3% |
7% |
3% |
Adverse Events Reported in Controlled Trials
|
Table 3. Adverse Events Reported in Controlled Clinical Trials in Mild to Moderate Alzheimer's Disease in at Least 2% of Patients Receiving donepezil hydrochloride and at a Higher Frequency than Placebo treated Patients |
||
|
Body System/Adverse Event
|
Placebo (n=355) |
Donepezil hydrochloride (n=747) |
|
Percent of Patients with any Adverse Event
|
72 |
74 |
|
Body as a Whole
|
|
|
| Headache |
9 |
10 |
| Pain, various locations |
8 |
9 |
| Accident |
6 |
7 |
| Fatigue |
3 |
5 |
|
Cardiovascular System
|
|
|
| Syncope |
1 |
2 |
|
Digestive System
|
|
|
| Nausea |
6 |
11 |
| Diarrhea |
5 |
10 |
| Vomiting |
3 |
5 |
| Anorexia |
2 |
4 |
|
Hemic and Lymphatic System
|
|
|
| Ecchymosis |
3 |
4 |
|
Metabolic and Nutritional Systems
|
|
|
| Weight Decrease |
1 |
3 |
|
Musculoskeletal System
|
|
|
| Muscle Cramps |
2 |
6 |
| Arthritis |
1 |
2 |
|
Nervous System
|
|
|
| Insomnia |
6 |
9 |
| Dizziness |
6 |
8 |
| Depression |
<1 |
3 |
| Abnormal Dreams |
0 |
3 |
| Somnolence |
<1 |
2 |
|
Urogenital System
|
|
|
| Frequent Urination |
1 |
2 |
Other Adverse Events Observed During Clinical Trials
Frequent adverse eventsInfrequent adverse events
Body as a Whole: Frequent:Infrequent:
Cardiovascular System: Frequent:Infrequent:
Digestive System: Frequent:Infrequent:
Endocrine System: Infrequent:
Hemic and Lymphatic System: Infrequent:
Metabolic and Nutritional Disorders: Frequent: Infrequent:
Musculoskeletal System: Frequent: Infrequent:
Nervous System: Frequent:Infrequent:
Respiratory System: Frequent:Infrequent:
Skin and Appendages: Frequent:Infrequent:
Special Senses: Frequent:Infrequent:
Urogenital System: Frequent:Infrequent:
Severe Alzheimer's Disease
Adverse Events Leading to Discontinuation
Most Frequent Adverse Clinical Events Seen in Association with the Use of donepezil hydrochloride
Adverse Events Reported in Controlled Trials
|
Table 4. Adverse Events Reported in Controlled Clinical Trials in Severe Alzheimer's Disease in at Least 2% of Patients Receiving Donepezil hydrochloride and at a Higher Frequency than Placebo treated Patients |
||
|
Body System/Adverse Event |
Placebo (n=392) |
Donepezil hydrochloride (n=501) |
|
Percent of Patients with any Adverse Event
|
73 |
81 |
|
Body as a Whole
|
|
|
| Accident |
12 |
13 |
| Infection |
9 |
11 |
| Headache |
3 |
4 |
| Pain |
2 |
3 |
| Back Pain |
2 |
3 |
| Fever |
1 |
2 |
| Chest Pain |
<1 |
2 |
|
Cardiovascular System
|
|
|
| Hypertension |
2 |
3 |
| Hemorrhage |
1 |
2 |
| Syncope |
1 |
2 |
|
Digestive System
|
|
|
| Diarrhea |
4 |
10 |
| Vomiting |
4 |
8 |
| Anorexia |
4 |
8 |
| Nausea |
2 |
6 |
|
Hemic and Lymphatic System |
|
|
| Ecchymosis |
2 |
5 |
|
Metabolic and Nutritional Systems
|
|
|
|
Creatine Phosphokinase Increased |
1 |
3 |
| Dehydration |
1 |
2 |
| Hyperlipemia |
<1 |
2 |
|
Nervous System
|
|
|
| Insomnia |
4 |
5 |
| Hostility |
2 |
3 |
| Nervousness |
2 |
3 |
| Hallucinations |
1 |
3 |
| Somnolence |
1 |
2 |
| Dizziness |
1 |
2 |
| Depression |
1 |
2 |
| Confusion |
1 |
2 |
| Emotional Lability |
1 |
2 |
| Personality Disorder |
1 |
2 |
|
Skin And Appendages
|
|
|
| Eczema |
2 |
3 |
|
Urogenital System
|
|
|
| Urinary Incontinence |
1 |
2 |
Other Adverse Events Observed During Clinical Trials
Frequent adverse eventsInfrequent adverse events
Body as a Whole: Frequent:Infrequent:
Cardiovascular System: Frequent: Infrequent:
Digestive System: Frequent:Infrequent:
Endocrine System: Infrequent:
Hemic and Lymphatic System: Frequent: Infrequent
Metabolic and Nutritional Disorders: Frequent: Infrequent:12
Musculoskeletal System: Frequent: Infrequent:
Nervous System: Frequent:Infrequent:
Respiratory System: Frequent:Infrequent:
Skin and Appendages: Frequent:Infrequent:
Special Senses: Infrequent:
Urogenital System: Frequent:Infrequent:
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on the Metabolism of Donepezil Hydrochloride
in vitro0-24max
7.2 Use with Anticholinergics
7.3 Use with Cholinomimetics and Other Cholinesterase Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C:8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
Because strategies for the management of overdose are continually evolving, it is advisable to contact a PoisonControlCenter to determine the latest recommendations for the management of an overdose of any drug.
11 DESCRIPTION
24293
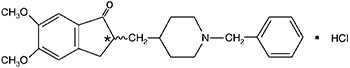
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
Donepezil Hydrochloride ODT 5 mg and 10 mg are bioequivalent to Donepezil Hydrochloride 5 mg and 10 mg tablets, respectively. A food effect study has not been conducted with Donepezil Hydrochloride ODT; however, the effect of food with Donepezil Hydrochloride ODT is expected to be minimal. Donepezil Hydrochloride ODT can be taken without regard to meals.
The elimination half life of donepezil is about 70 hours, and the mean apparent plasma clearance (Cl/F) is 0.13 – 0.19 L/hr/kg. Following multiple dose administration, donepezil accumulates in plasma by 4-7 fold, and steady state is reached within 15 days. The steady state volume of distribution is 12 - 16 L/kg. Donepezil is approximately 96% bound to human plasma proteins, mainly to albumins (about 75%) and alpha1 – acid glycoprotein (about 21%) over the concentration range of 2-1000 ng/mL.
Donepezil is both excreted in the urine intact and extensively metabolized to four major metabolites, two of which are known to be active, and a number of minor metabolites, not all of which have been identified.
Donepezil is metabolized by CYP 450 isoenzymes 2D6 and 3A4 and undergoes glucuronidation. Following administration of 14C-labeled donepezil, plasma radioactivity, expressed as a percent of the administered dose, was present primarily as intact donepezil (53%) and as 6-O-desmethyl donepezil (11%), which has been reported to inhibit AChE to the same extent as donepezil in vitro and was found in plasma at concentrations equal to about 20% of donepezil. Approximately 57% and 15% of the total radioactivity was recovered in urine and feces, respectively, over a period of 10 days, while 28% remained unrecovered, with about 17% of the Donepezil dose recovered in the urine as unchanged drug. Examination of the effect of CYP2D6 genotype in Alzheimer’s patients showed differences in clearance values among CYP2D6 genotype subgroups. When compared to the extensive metabolizers, poor metabolizers had a 31.5% slower clearance and ultra-rapid metabolizers had a 24% faster clearance. These results suggest CYP2D6 has a minor role in the metabolism of donepezil.
Hepatic Disease: In a study of 10 patients with stable alcoholic cirrhosis, the clearance of Donepezil Hydrochloride was decreased by 20% relative to 10 healthy ageand sex-matched subjects.
Renal Disease: In a study of 11 patients with moderate to severe renal impairment (ClC < 18 mL/min/1.73 m2) the clearance of Donepezil Hydrochloride did not differ from 11 age- and sex-matched healthy subjects.
Age: No formal pharmacokinetic study was conducted to examine agerelated differences in the pharmacokinetics of Donepezil Hydrochloride. Population pharmacokinetic analysis suggested that the clearance of donepezil in patients decreases with increasing age. When compared with 65-year old, subjects, 90-year old subjects have a 17% decrease in clearance, while 40- year old subjects have a 33% increase in clearance. The effect of age on donepezil clearance may not be clinically significant.
Gender and Race: No specific pharmacokinetic study was conducted to investigate the effects of gender and race on the disposition of Donepezil Hydrochloride. However, retrospective pharmacokinetic analysis and population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer’s disease indicates that gender and race (Japanese and Caucasians) did not affect the clearance of Donepezil Hydrochloride to an important degree.
Body weight: There was a relationship noted between body weight and clearance. Over the range of body weight from 50 kg to 110 kg, clearance increased from 7.77 L/h to 14.04 L/h, with a value of 10 L/hr for 70 kg individuals.
Drug Interactions: Effect of Donepezil Hydrochloride on the Metabolism of Other Drugs
No in vivo clinical trials have investigated the effect of Donepezil Hydrochloride on the clearance of drugs metabolized by CYP 3A4 (e.g. cisapride, terfenadine) or by CYP 2D6 (e.g. imipramine). However, in vitro studies show a low rate of binding to these enzymes (mean Ki about 50-130 µM), that, given the therapeutic plasma concentrations of donepezil (164 nM), indicates little likelihood of interference. Based on in vitro studies, donepezil shows little or no evidence of direct inhibition of CYP2B6, CYP2C8 and CYP2C19 at clinically relevant concentrations.
Whether Donepezil Hydrochloride has any potential for enzyme induction is not known. Formal pharmacokinetic studies evaluated the potential of Donepezil Hydrochloride for interaction with theophylline, cimetidine, warfarin, digoxin and ketoconazole. No effects of Donepezil Hydrochloride on the pharmacokinetics of these drugs were observed.
Effect of Other Drugs on the Metabolism of Donepezil Hydrochloride
A small effect of CYP2D6 inhibitors was identified in a population pharmacokinetic analysis of plasma donepezil concentrations measured in patients with Alzheimer’s disease. Donepezil clearance was reduced by approximately 17% in patients taking 10 or 23 mg in combination with a known CYP2D6 inhibitor. This result is consistent with the conclusion that CYP2D6 is a minor metabolic pathway of donepezil.
Formal pharmacokinetic studies demonstrated that the metabolism of Donepezil Hydrochloride is not significantly affected by concurrent administration of digoxin or cimetidine. An in vitro study showed that donepezil was not a substrate of P-glycoprotein.
Drugs Highly Bound to Plasma Proteins Drug displacement studies have been performed in vitro between this highly bound drug (96%) and other drugs such as furosemide, digoxin, and warfarin. Donepezil hydrochloride at concentrations of 0.3-10 micrograms/mL did not affect the binding of furosemide (5 micrograms/mL), digoxin (2 ng/mL), and warfarin (3 micrograms/mL) to human albumin. Similarly, the binding of donepezil hydrochloride to human albumin was not affected by furosemide, digoxin and warfarin.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
13.2 AnimalToxicology
14 CLINICAL STUDIES
.
14.1 Mild to Moderate Alzheimer's disease
Study Outcome Measures:
Thirty-Week Study
Effects on the ADAS-cog:
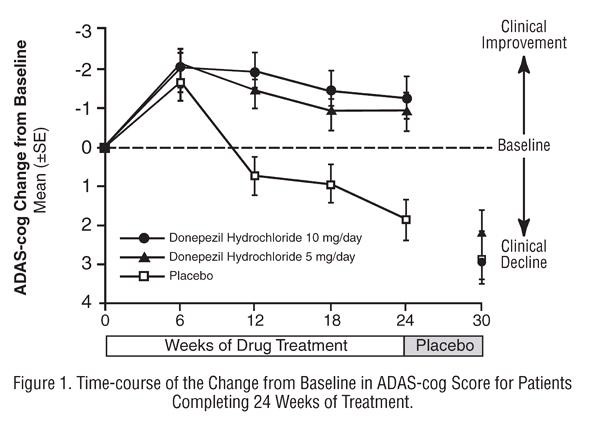
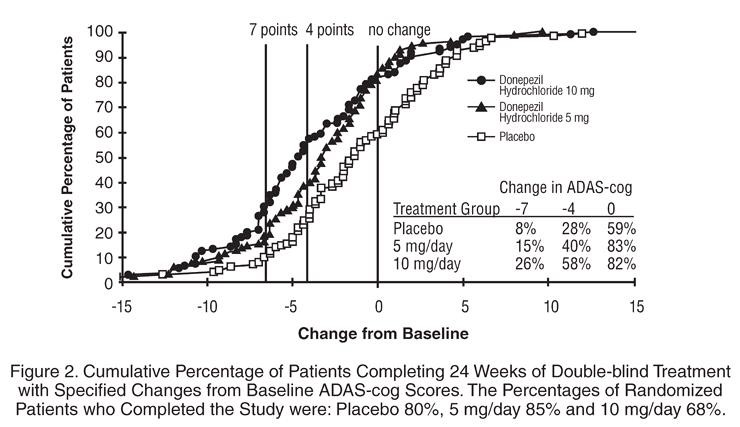
Effects on the CIBIC-plus:

Fifteen-Week Study
Effects on the ADAS-Cog:
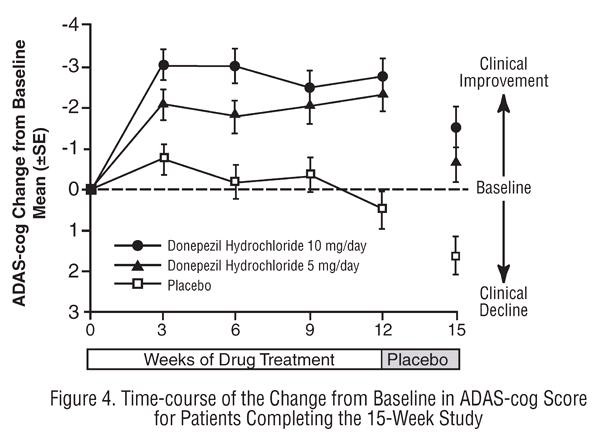
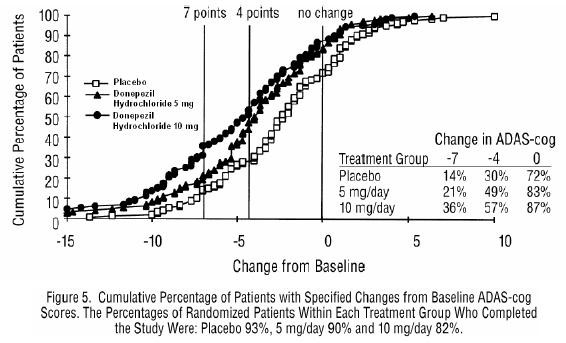
Effects on the CIBIC-plus:
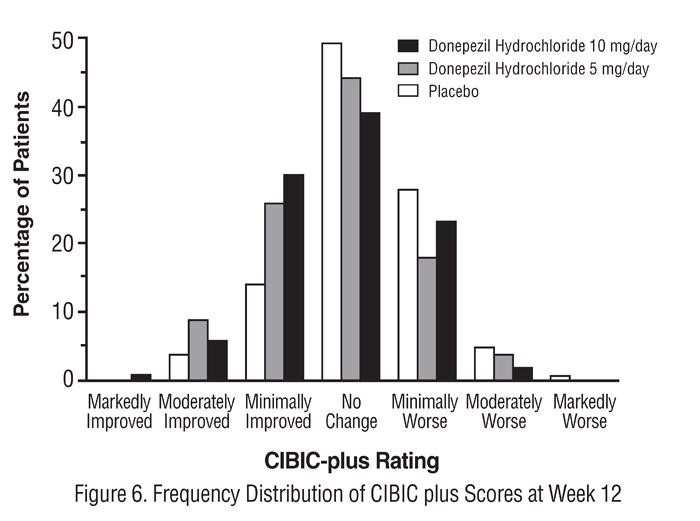
14.2 Moderate to Severe Alzheimer's Disease
The effectiveness of donepezil hydrochloride in the treatment of patients with moderate to severe Alzheimer’s Disease was established in studies employing doses of 10 mg/day.
Swedish 6 Month Study
Study Outcome Measures:
Effects on the SIB:
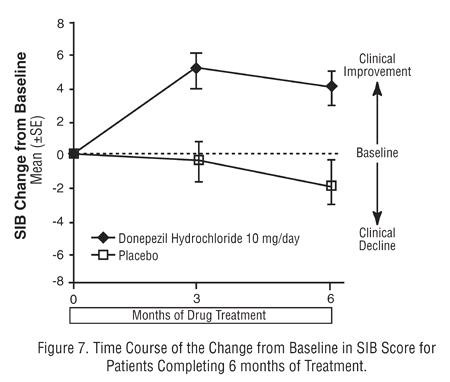
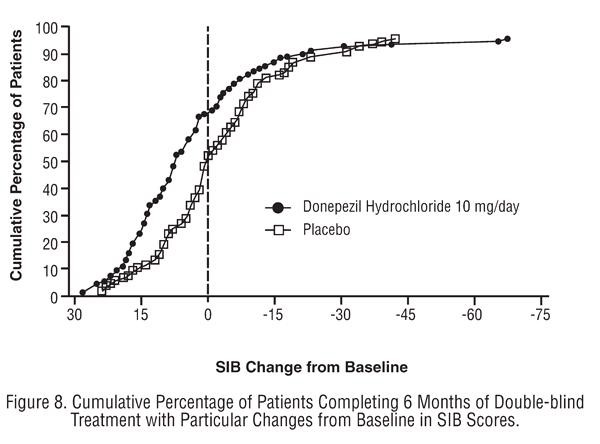

Effects on the ADCS-ADL-severe
Figure 10
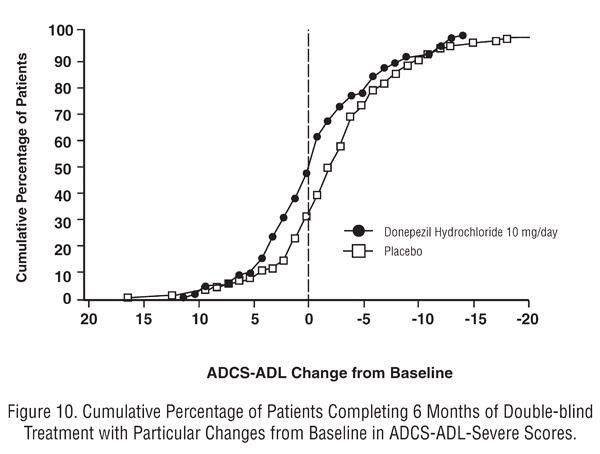
Japanese 24-Week Study
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 Donepezil Hyrochloride Tablets USP
Supplied as round tablets containing either 5mg or 10mg of donepezil hydrochloride USP.
The 5 mg orally disintegrating tablets USP are yellow coloured, circular, flat face beveled edge uncoated tablets dedossed with “CL 31” on one side and plain on the other side.
5 mg Unit Dose Blister Package 30 (10x3)
(NDC 33342-029-06)
5 mg Unit Dose Blister Package 100 (10x10)
(NDC 33342-029-12)
5 mg Container Pack of 30’s
(NDC 33342-029-07)
The 10 mg orally disintegrating tablets USP are yellow coloured, circular, flat face beveled edge uncoated tablets dedossed with “CL 32” on one side and plain on the other side.
10 mg Unit Dose Blister Package 30 (10x3)
(NDC 33342-030-06)
10 mg Unit Dose Blister Package 100 (10x10)
(NDC 33342-030-12)
10 mg Container Pack of 30’s
(NDC 33342-030-07)
Storage:
17 PATIENT COUNSELING INFORMATION
PATIENT PACKAGE INSERT
Donepezil hydrochloride ODT USP : 5mg and 10mg
What are donepezil hydrochloride orally disintegrating tablets (ODT)?
- Seem much better
- Get better in small ways or stay the same
- Get worse over time but slower than expected
- Not change and get worse as expected
Who should not take donepezil hydrochloride ODT?
What should I tell the doctor before the patent takes donepezil hydrochloride ODT?
- Any heart problems including problems with irregular, slow, or fast heartbeats
- Asthama or lung problems
- A seizure
- Stomach ulcers
- Difficulty passing urine
- Liver or kidney problems
- Trouble swallowing tablets
- Present pregnancy or plans to become pregnant. It is not known if donepezil hydrochloride can harm an unborn baby.
- Present breast-feeding. It is not known if donepezil hydrochloride passes into breast milk. Donepezil hydrochloride ODT is not for women who are breast-feeding.
Tell the doctor about all medicines the patient takes,
- Surgery
- medical procedures
- dental surgery or procedures
How should the patient take donepezil hydrochloride ODT?
- Give donepezil hydrochloride ODT exactly as prescribed by the doctor. Do not stop donepezil hydrochloride ODT or change the dose yourself. Talk with the doctor first.
- Give donepezil hydrochloride ODT one time each day. Donepezil hydrochloride ODT can be taken with or without food.
- Donepezil hydrochloride ODT melts on the tongue. The patient should drink some water after the tablet melts.
- If you miss giving the patient a dose of donepezil hydrochloride ODT, just wait. Give only the next dose at the usual time. Do not give 2 doses at the same time.
- If donepezil hydrochloride ODT is missed for 7 days or more, talk with the doctor before starting again.
- If the patient takes too much donepezil hydrochloride at one time, call the doctor or poison control center, or go to the emergency room right away.
Donepezil hydrochloride ODT may cause the following serious side effects:
- slow heartbeat and fainting. This happens more often in people with heart problems. Call the doctor right away if the patient faints while taking donepezil hydrochloride ODT.
- more stomach acid: This raises the chance of ulcers and bleeding. The risk is higher for patients who had ulcers, or take aspirin or other NSAIDs.
- worsening of lung problem in people with asthma or other lung diseases.
- siezures.
- difficulty passing urine.
- fainting.
- heartburn or stomach pain that is new or won't go away.
- nausea or vomiting, blood in the vomit, dark vomit that looks like coffee grounds.
- bowel movement or stools that look like black tar.
- new or worse asthma or breathing problem.
- seizures.
- difficulty passing urine.
- nausea
- diarrhea not sleeping well
- vomiting
- muscle cramps
- feeling tired
- not wanting to eat
How should Donepezil hydrochloride ODT be stored?
Keep donepezil hydrochloride ODT and all medicines out of the reach of children.
What are the ingredients in donepezil hydrochloride ODT?
Active ingredient:
Inactive Ingredients:
Manufactured for:
Macleods Pharma USA, INC,
Plainsboro, NJ 08536
Manufactured by:
Macleods Pharmaceutical Ltd.
Nalgarh, Himachal Pradesh, India.
January2013
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Rx only
30 TABLETS
Donepezil Hydrochloride Orally Disintegrating Tablets USP

NDC 33342-030-07
Rx only
30 TABLETS
Donepezil Hydrochloride Orally Disintegrating Tablets USP

Donepezil HydrochlorideDonepezil Hydrochloride TABLET, ORALLY DISINTEGRATING
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Donepezil HydrochlorideDonepezil Hydrochloride TABLET, ORALLY DISINTEGRATING
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||