Diurex
Alva-Amco Pharmacal Companies, Inc.
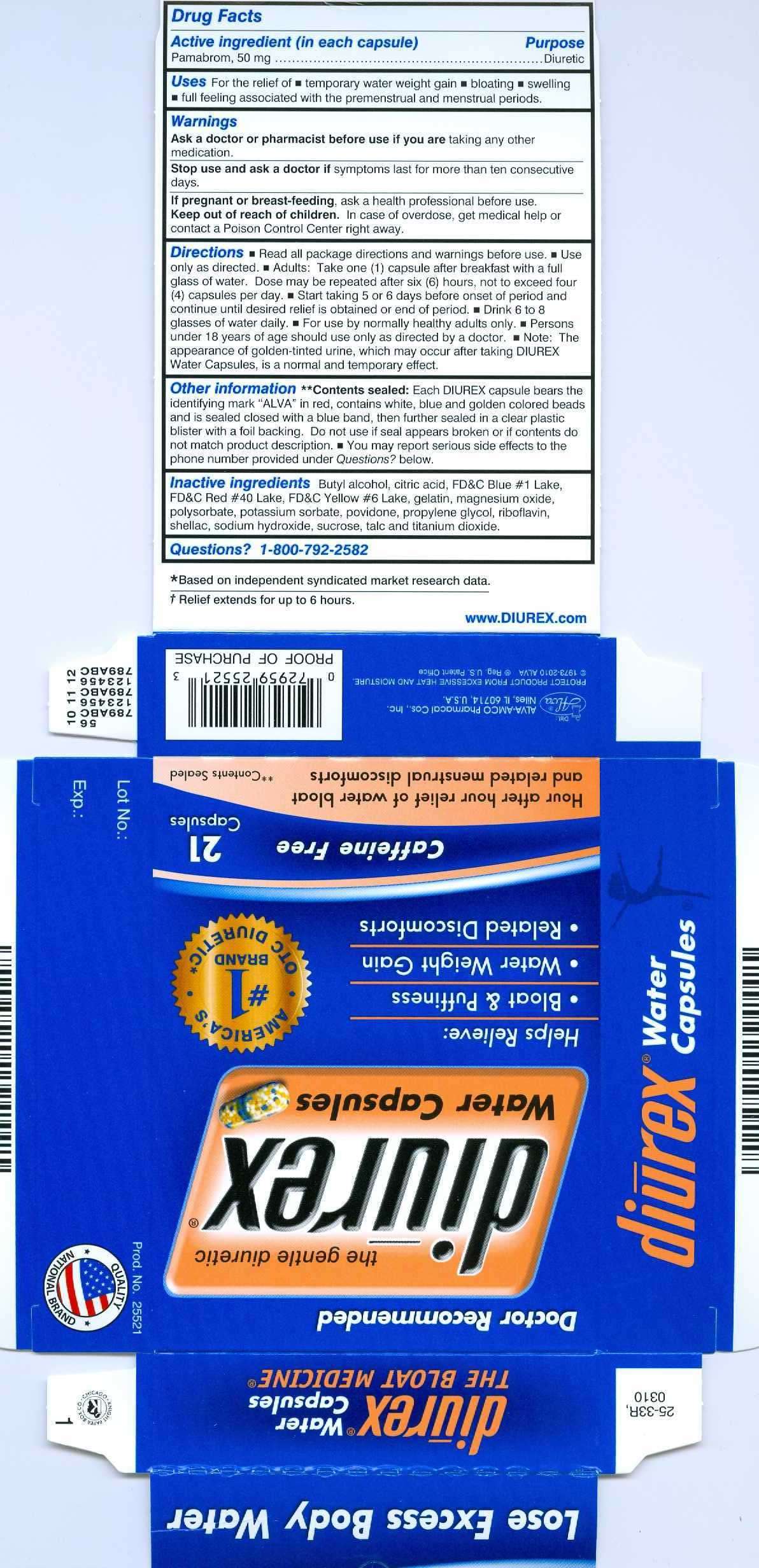
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient (in each capsule)
- Diurex Uses
- Warnings
- Directions
- Other Information
- Inactive ingredients
FULL PRESCRIBING INFORMATION
Active ingredient (in each capsule)
Pamabrom, 50 mg.........................................Diuretic
Diurex Uses
For the relief of
- temporary water weight gain
- bloating
- swelling
- full feeling
Warnings
Ask a doctor or pharmacist before use if you are taking any other medication.
Stop use and ask a doctor if symptoms last for more than ten consecutive days.
If pregnant or breastfeeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- Read all package directions and warnings before use.
- Use only as directed.
- Adults: Take one (1) capsule after breakfast with a full glass of water. Dose may be repeated after six (6) hours, not to exceed four (4) capsules per day.
- Start taking 5 or 6 days before onset of period and continue until desired relief is obtained or end of period.
- Drink 6 to 8 glasses of water daily.
- For use by normally healthy adults only.
- Persons under 18 years of age should use only as directed by a doctor.
- Note: The appearance of golden-tinted urine, which may occur after taking Diurex Water capsules, is a normal and temporary effect.
Other Information
**Contents sealed: Each Diurex Capsule bears the identifying mark "ALVA" in red, contains white, blue and golden colored beads and is sealed closed with a blue band, then further sealed in a clear plastic blister with a foil backing. Do not use if seal appears broken or if contents do not match product description. You may report serious side effects to the phone number provided under Questions? below.
Inactive ingredients
Butyl alcohol, citric acid, FDC Blue #1 Lake, FDC Red #40 Lake, FDC Yellow #6 Lake, gelatin, magnesium oxide, polysorbate, potassium sorbate, povidone, propylene glycol, riboflavin, shellac, sodium hydroxide, sucrose, talc and titanium dioxide.
Questions? 1-800-792-2582
DiurexPamabrom CAPSULE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||