Daytime Cold and Pain Relief
Safeway 44-503
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredients (in each caplet)
- Purpose
- Daytime Cold and Pain Relief Uses
- Warnings
- Directions
- Daytime Cold and Pain Relief Other information
- Inactive ingredients
- Principal Display Panel
FULL PRESCRIBING INFORMATION
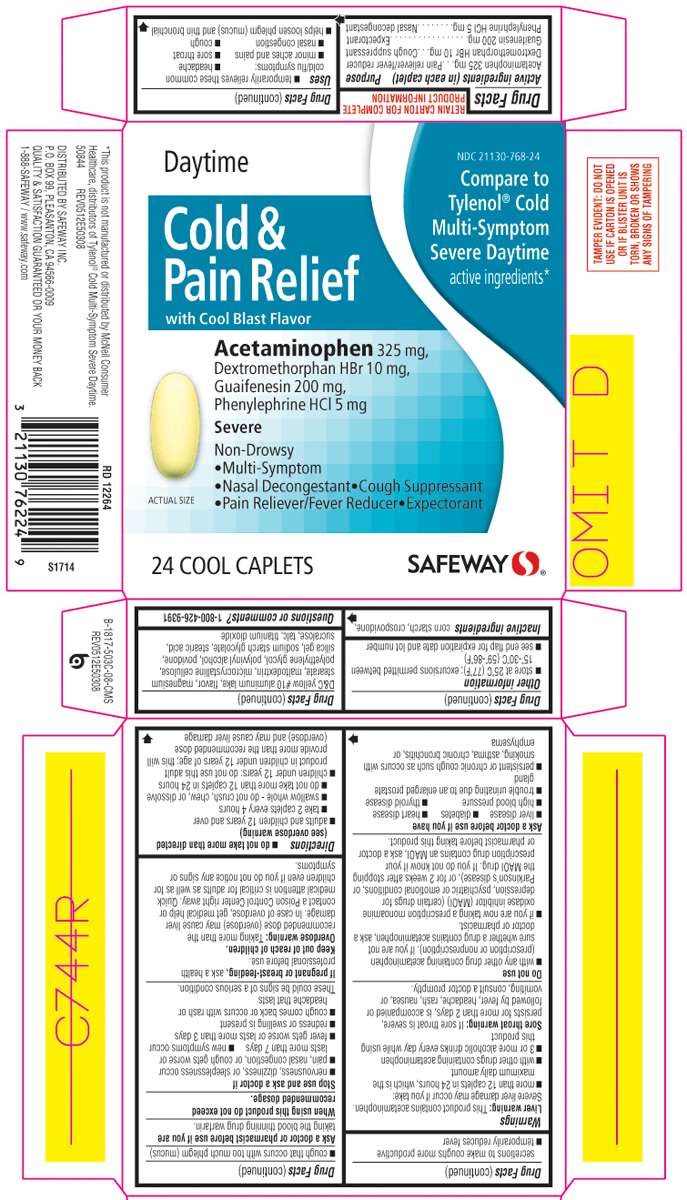
Active ingredients (in each caplet)
Acetaminophen 325 mg
Dextromethorphan HBr 10 mg
Guaifenesin 200 mg
Phenyleprhine HCl 5 mg
Purpose
Pain reliever/fever reducer
Cough suppressant
Expectorant
Nasal decongestant
Daytime Cold and Pain Relief Uses
- temporarily relieves these common cold/flu symptoms
- minor aches and pains
- headache
- nasal congestion
- sore throat
- cough
- helps loosen phlegm (mucus) and thin bronchial secretions to make coughs more productive
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 12 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Sore throat warning: if sore throat is severe, persists for more than 2 days, is accompanied for followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- trouble urinating due to an enlarged prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough that occurs with too much phlegm (mucus)
Ask a doctor or pharmacist before use if you are
taking the blood thinning drug warfarin.
When using this product
do not exceed recommended dosage.
Stop use and ask a doctor if
- nervousness, dizziness, or sleeplessness occur
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- new symptoms occur
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
Overdose warning: Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
- do not take more than directed (see overdose warning)
- adults and children 12 years and over
- take 2 caplets every 4 hours
- swallow whole - do not crush, chew, or dissolve
- do not take more than 12 caplets in 24 hours
- children under 12 years: do not use this adult product in children under 12 years of age; this will provide more than the recommended dose (overdose) and may cause liver damage
Daytime Cold and Pain Relief Other information
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, crospovidone, D&C yellow #10 aluminum lake, flavor, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silica gel, sodium starch glycolate, stearic acid, sucralose, talc, titanium dioxide
Principal Display Panel
NDC 21130-503-08
Compare to Tylenol ® Cold Multi-Symptom Severe Daytime active ingredients*
Day Time
Cold &
Pain Relief
with Cool Blast Flavor
Acetaminophen 325 mg,
Dextromethorphan HBr 10 mg,
Guaifenesin 200 mg,
Phenylephrine HCl 5 mg
Severe
Non-Drowsy
• Multi-Symptom
• Nasal Decongestant • Cough Suppressant
• Pain Reliever/Fever Reducer • Expectorant
24 COOL CAPLETS
SAFEWAY™
*This product is not manufactured or distributed by McNeil Consumer Healthcare, owner of the registered trademark Tylenol® Cold Head Congestion Severe Daytime.
50844 REV0512E50308
DISTRIBUTED BY SAFEWAY INC., P.O. BOX 99, PLEASANTON, CA 94566-009
QUALITY & SATISFACTION GUARANTEED OR YOUR MONEY BACK
1-888-SAFEWAY / www.safeway.com
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
Daytime Cold and Pain ReliefAcetaminophen, Phenylephrine HCl, Dextromethorphan HBr and Guiafenesin TABLET, COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||