Aluvea
ALUVEA
FULL PRESCRIBING INFORMATION: CONTENTS*
- ALUVEA DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATION AND USAGE
- ALUVEA CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- ALUVEA ADVERSE REACTIONS
- ALUVEA DOSAGE AND ADMINISTRATIONS
- KEEP THIS AND ALL OTHER MEDICATIONS OUT OF REACH OF CHILDREN
- HOW SUPPLIED
- PRINCIPAL DISPLAY PANEL - 227g Carton
FULL PRESCRIBING INFORMATION
(39% Urea Cream)
RX ONLY
ALUVEA DESCRIPTION
Aluvea™ (urea) cream, 39% is a keratolytic emollient, which is a gentle, yet effective, tissue softener for skin.
Each gram of Aluvea Cream contains 39% Urea as the active ingredient, and the following inactive ingredients: dimethyl isosorbide, emulsifying wax NF, glycerin 99.7% USP, isopropyl myristate, purified water, sorbitol 70% Solution USP, tridecyl stearate and neopentyl glycol dicaprylate/dicaprate and tridecyl trimellitate.
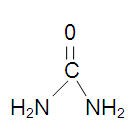
CHEMICAL STRUCTURE
Urea is diamide of Carbonic acid with the following structure:
CLINICAL PHARMACOLOGY
Urea gently dissolves the intracellular matrix which results in loosening of the horny layer of the skin and shedding of scaly skin at regular intervals, thereby softening hyperkeratotic areas of the skin.
PHARMACOKINETICS
The exact mechanism of action of topically applied urea is not known.
INDICATION AND USAGE
Urea is useful for the treatment of hyperkeratotic conditions such as dry, rough skin, xerosis, ichthyosis, skin cracks and fissures, dermatitis, eczema, psoriasis, keratoses and calluses.
ALUVEA CONTRAINDICATIONS
Known hypersensitivity to any of the listed ingredients
WARNINGS
For external use only. Avoid contact with eyes, lips or mucous membranes.
PRECAUTIONS
Urea should be used as directed by a physician and should not be used to treat conditions other than those for which it was prescribed. If redness or irritation occurs, discontinue use.
Pregnancy
Category C
Animal reproduction studies have not been conducted with Aluvea. It is also not known whether Aluvea can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. Aluvea should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether Aluvea is excreted in human milk, therefore caution should be exercised when administering to a nursing mother.
ALUVEA ADVERSE REACTIONS
Transient stinging, burning, itching or irritation is possible and normally resolves upon discontinuing the medication.
DOSAGE AND ADMINISTRATIONS
Apply Aluvea to affected skin twice per day, or as directed by your physician. Rub in until completely absorbed.
KEEP THIS AND ALL OTHER MEDICATIONS OUT OF REACH OF CHILDREN
HOW SUPPLIED
Aluvea is supplied in an 8oz. bottle.
NDC 0259-1139-80
Store at room temperature 15°C -30°C (59°F-86°F).
Protect from freezing.
Manufactured for:
Merz Pharmaceuticals, LLC
Greensboro, NC 27410
5011457 12/10
PRINCIPAL DISPLAY PANEL - 227g Carton
NDC 0259-1139-80
ALUVEA
™
39% Urea Cream
WITH UNIQUE
CSP ™
VEHICLE
TECHNOLOGY
FOR EXTERNAL USE ONLY
8 oz. (227g)
Rx ONLY

AluveaUrea CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||