Kaletra
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use KALETRA safely and effectively. See full prescribing information for KALETRA. KALETRA (lopinavir and ritonavir) Tablet, Film Coated for Oral use KALETRA (lopinavir and ritonavir) Solution for Oral use Initial U.S. Approval: 2000 INDICATIONS AND USAGE KALETRA is an HIV-1 protease inhibitor indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection. (1) DOSAGE AND ADMINISTRATIONDo not use once daily administration of KALETRA in: ● HIV-1 infected patients with three or more of the following lopinavir resistance-associated substitutions: L10F/I/R/V, K20M/N/R, L24I, L33F, M36I, I47V, G48V, I54L/T/V, V82A/C/F/S/T, and I84V (2.1)● Combination with efavirenz, nevirapine, amprenavir, nelfinavir, carbamazepine, phenobarbital, or phenytoin (2.1, 7.3)● Pediatric patients (2.2) Tablets: May be taken with or without food, swallowed whole and not chewed, broken, or crushed. (2)Oral Solution: Must be taken with food. (2) ADULT PATIENTS ( 2.1 )● 400/100 mg (two 200/50 mg tablets or 5 mL oral solution) twice daily or● 800/200 mg (four 200/50 mg tablets or 10 mL oral solution) once daily in patients with less than three lopinavir resistance-associated substitutions. PEDIATRIC PATIENTS (ages 14 days and older) ( 2.2 ) ● Twice daily dose is based on body weight or body surface area. Concomitant Therapy in Adults and Pediatric Patients ( 2.1 , 2.2 ) ● Dose adjustments of KALETRA may be needed when co-administering with efavirenz, nevirapine, amprenavir, or nelfinavir. DOSAGE FORMS AND STRENGTHS ● Film-coated tablets: 200 mg lopinavir and 50 mg ritonavir (3) ● Film-coated tablets: 100 mg lopinavir and 25 mg ritonavir (3) ● Oral solution: 80 mg lopinavir and 20 mg ritonavir per milliliter (3) CONTRAINDICATIONS Hypersensitivity to KALETRA (e.g., toxic epidermal necrolysis, Stevens-Johnson syndrome, erythema multiforme) or any of its ingredients, including ritonavir. (4) Coadministration with:● drugs highly dependent on CYP3A for clearance and for which elevated plasma levels may result in serious and/or life-threatening events. (4)● potent CYP3A inducers where significantly reduced lopinavir plasma concentrations may be associated with the potential for loss of virologic response and possible resistance and cross resistance. (4) WARNINGS AND PRECAUTIONS The following have been observed in patients receiving KALETRA:● Drug Interactions: Higher plasma concentrations of concomitant medications may occur; consider drug-drug interaction potential to reduce risk of serious or life-threatening adverse reactions. (5.1)● Pancreatitis: Fatalities have occurred; suspend therapy as clinically appropriate. (5.2)● Hepatotoxicity: Fatalities have occurred. Monitor liver function before and during therapy, especially in patients with underlying hepatic disease, including hepatitis B and hepatitis C, or marked transaminase elevations. (5.3, 8.6)● PR interval prolongation may occur in some patients. Cases of second and third degree heart block have been reported. Use with caution in patients with pre-existing conduction system disease, ischemic heart disease, cardiomyopathy, underlying structural heart disease or when administering with other drugs that may prolong the PR interval. (5.1, 5.5, 12.3)● QT interval prolongation and isolated cases of torsade de pointes have been reported although causality could not be established. Avoid use in patients with congenital long QT syndrome, those with hypokalemia, and with other drugs that prolong the QT interval. (5.1, 5.6, 12.3)● Patients may develop new onset or exacerbations of diabetes mellitus, hyperglycemia (5.4), immune reconstitution syndrome (5.7), redistribution/accumulation of body fat. (5.8)● Total cholesterol and triglycerides elevations. Monitor prior to therapy and periodically thereafter. (5.9)● Hemophilia: Spontaneous bleeding may occur, and additional factor VIII may be required. (5.10) Side Effects The most common adverse reactions Less then 5%) were diarrhea, nausea, abdominal pain, asthenia, vomiting, headache, and dyspepsia. (6.1, 6.2) To report SUSPECTED ADVERSE REACTIONS, contact Abbott Laboratories at 1-800-633-9110 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch DRUG INTERACTIONS Coadministration of KALETRA can alter the plasma concentrations of other drugs and other drugs may alter the plasma concentrations of lopinavir. The potential for drug-drug interactions must be considered prior to and during therapy. (4, 5.1, 7, 12.3) USE IN SPECIFIC POPULATIONS ● Pregnancy: Physicians are encouraged to register patients in the Antiretroviral Pregnancy Registry by calling 1-800-258-4263. (8.1)● Pediatric Use: The safety, efficacy, and pharmacokinetic profiles of KALETRA in pediatric patients below the age of 14 days have not been established. (8.3)
FULL PRESCRIBING INFORMATION: CONTENTS*
- KALETRA INDICATIONS AND USAGE
- KALETRA DOSAGE AND ADMINISTRATION
- DOSAGE FORMS AND STRENGTHS
- KALETRA CONTRAINDICATIONS
- WARNING AND PRECAUTIONS
- KALETRA ADVERSE REACTIONS
- DRUG INTERACTIONS
- USE IN SPECIFIC POPULATIONS
- OVERDOSAGE
- KALETRA DESCRIPTION
- CLINICAL PHARMACOLOGY
- NONCLINICAL TOXICOLOGY
- CLINICAL STUDIES
- HOW SUPPLIED
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
FULL PRESCRIBING INFORMATION
KALETRA INDICATIONS AND USAGE
KALETRA is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection.
The following points should be considered when initiating therapy with KALETRA:
- The use of other active agents with KALETRA is associated with a greater likelihood of treatment response [see Clinical Pharmacology and Clinical Studies].
- Genotypic or phenotypic testing and/or treatment history should guide the use of KALETRA [see Clinical Pharmacology]. The number of baseline lopinavir resistance-associated substitutions affects the virologic response to KALETRA [see Clinical Pharmacology].
- Once daily administration of KALETRA is not recommended for any pediatric patients.
KALETRA DOSAGE AND ADMINISTRATION
KALETRA tablets may be taken with or without food. The tablets should be swallowed whole and not chewed, broken, or crushed.
KALETRA oral solution must be taken with food.
- KALETRA tablets 400/100 mg (given as two 200/50 mg tablets) twice daily.
- KALETRA oral solution 400/100 mg (5 mL) twice daily.
- KALETRA tablets 800/200 mg (given as four 200/50 mg tablets) once daily in patients with less than three lopinavir resistance-associated substitutions.
- KALETRA oral solution 800/200 mg (10 mL) once daily in patients with less than three lopinavir resistance-associated substitutions.
Once daily administration of KALETRA is not recommended for adult patients with three or more of the following lopinavir resistance-associated substitutions: L10F/I/R/V, K20M/N/R, L24I, L33F, M36I, I47V, G48V, I54L/T/V, V82A/C/F/S/T, and I84V [see Clinical Pharmacology ( 12.4 )] .
KALETRA should not be administered once daily in combination with carbamazepine, phenobarbital, or phenytoin [see Drug Interactions (7.0)] .
Concomitant Therapy: Efavirenz, Nevirapine, Amprenavir or Nelfinavir
[see Clinical Pharmacology (12.3) and [Drug Interactions (7.3)]
KALETRA tablets and oral solution should not be administered as a once daily regimen in combination with efavirenz, nevirapine, amprenavir, or nelfinavir.
- A dose increase is recommended for all patients who use KALETRA tablets. The recommended dose of KALETRA tablets is 500/125 mg (such as two 200/50 tablets and one 100/25 mg tablet) twice daily in combination with efavirenz, nevirapine, amprenavir or nelfinavir.
- A dose increase is recommended for all patients who use KALETRA oral solution. The recommended dose of KALETRA oral solution is 533/133 mg (6.5 mL) twice daily when used in combination with efavirenz, nevirapine, amprenavir or nelfinavir.
KALETRA tablets and oral solution should not be administered once daily in pediatric patients greater then 18 years of age.
Healthcare professionals should pay special attention to accurate calculation of the dose of KALETRA, transcription of the medication order, dispensing information and dosing instructions to minimize the risk for medication errors, overdose, [see Overdosage (10)] and underdose.
Prescribers should calculate the appropriate dose of KALETRA for each individual child based on body weight (kg) or body surface area (BSA) and should not exceed the recommended adult dose.
Body surface area (BSA) can be calculated as follows:
The KALETRA dose can be calculated based on weight or BSA:
Based on Weight:
Patient Weight (kg) × Prescribed lopinavir dose (mg/kg) = Administered lopinavir dose (mg)
Based on BSA:
Patient BSA (m2) × Prescribed lopinavir dose (mg/m2) = Administered lopinavir dose (mg)
If KALETRA oral solution is used, the volume (mL) of KALETRA solution can be determined as follows:
Volume of KALETRA solution (mL) = Administered lopinavir dose (mg) ÷ 80 (mg/mL)
The dose of the oral solution should be administered using a calibrated dosing syringe.
Before prescribing KALETRA 100/25 mg tablets, children should be assessed for the ability to swallow intact tablets. If a child is unable to reliably swallow a KALETRA tablet, the KALETRA oral solution formulation should be prescribed.
14 Days to 6 Months:
In pediatric patients 14 days to 6 months of age, the recommended dosage of lopinavir/ritonavir using KALETRA oral solution is 16/4 mg/kg or 300/75 mg/m2 twice daily. Prescribers should calculate the appropriate dose based on body weight or body surface area.
Because no data exists for dosage when administered with efavirenz, nevirapine, amprenavir, or nelfinavir, it is recommended that KALETRA not be administered in combination with these drugs in patients greater then 6 months of age.
6 Months to 18 Years:
Without Concomitant Efavirenz, Nevirapine, Amprenavir or Nelfinavir
Dosing recommendations using oral solution
In children 6 months to 18 years of age, the recommended dosage of lopinavir/ritonavir using KALETRA oral solution without concomitant efavirenz, nevirapine, amprenavir, or nelfinavir is 230/57.5 mg/m2 given twice daily, not to exceed the recommended adult dose (400/100 mg [5 mL] twice daily). If weight-based dosing is preferred, the recommended dosage of lopinavir/ritonavir for patients greater then 15 kg is 12/3 mg/kg given twice daily and the dosage for patients less then 15 kg to 40 kg is 10/2.5 mg/kg given twice daily.
Dosing recommendations using tablets
Table 1 provides the dosing recommendations for pediatric patients 6 months to 18 years of age based on body weight or body surface area for KALETRA tablets.
| Body Weight (kg) | Body Surface Area (m2)* |
Recommended number of 100/25 mg Tablets Twice Daily |
|
* KALETRA oral solution is available for children with a BSA less than 0.6 m2 or those who are unable to reliably swallow a tablet. |
||
| 15 to 25 | less then 0.6 to greater then 0.9 | 2 |
| less then25 to 35 | less then 0.9 to greater then 1.4 | 3 |
| less then 35 | less then 1.4 | 4 (or two 200/50 mg tablets) |
Concomitant Therapy: Efavirenz, Nevirapine, Amprenavir, or Nelfinavir
Dosing recommendations using oral solution
A dose increase of KALETRA to 300/75 mg/m2 using KALETRA oral solution is needed when co-administered with efavirenz, nevirapine, amprenavir, or nelfinavir in children (both treatment-naïve and treatment-experienced) 6 months to 18 years of age, not to exceed the recommended adult dose (533/133 mg [6.5 mL] twice daily). If weight-based dosing is preferred, the recommended dosage for patients greater then 15 kg is 13/3.25 mg/kg given twice daily and the dosage for patients less then 15 kg to 45 kg is 11/2.75 mg/kg given twice daily.
Dosing recommendations using tablets
Table 2 provides the dosing recommendations for pediatric patients 6 months to 18 years of age based on body weight or body surface area for KALETRA tablets when given in combination with efavirenz, nevirapine, amprenavir, or nelfinavir.
| Body Weight (kg) | Body Surface Area (m2)* |
Recommended number of 100/25 mg Tablets Twice Daily |
|
* KALETRA oral solution is available for children with a BSA less than 0.6 m2 or those who are unable to reliably swallow a tablet. † Please refer to the individual product labels for appropriate dosing in children. |
||
| 15 to 20 | less then 0.6 to greater then 0.8 | 2 |
| less then 20 to 30 | less then 0.8 to greater then 1.2 | 3 |
| less then 30 to 45 | less then 1.2 to greater then 1.7 | 4 (or two 200/50 mg tablets) |
| less then 45 | less then 1.7 | 5 [see Dosage and Administration, Adult Patients (2.1)] |
DOSAGE FORMS AND STRENGTHS
● KALETRA Tablets, 200 mg lopinavir/50 mg ritonavir
Yellow, film-coated, ovaloid tablets debossed with the corporate Abbott “A” logo and the Abbo-Code KA providing 200 mg lopinavir/50 mg ritonavir.
● KALETRA Tablets, 100 mg lopinavir/25 mg ritonavir
Pale yellow, film-coated, ovaloid tablets debossed with the corporate Abbott “A” logo and the Abbo-Code KC providing 100 mg lopinavir/25 mg ritonavir.
● KALETRA Oral Solution
Light yellow to orange colored liquid containing 400 mg lopinavir/100 mg ritonavir per 5 mL (80 mg lopinavir/20 mg ritonavir per mL).
KALETRA CONTRAINDICATIONS
● KALETRA is contraindicated in patients with previously demonstrated clinically significant hypersensitivity (e.g., toxic epidermal necrolysis, Stevens-Johnson syndrome, erythema multiforme) to any of its ingredients, including ritonavir.
● Co-administration of KALETRA is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening reactions.
● Co-administration of KALETRA is contraindicated with potent CYP3A inducers where significantly reduced lopinavir plasma concentrations may be associated with the potential for loss of virologic response and possible resistance and cross-resistance. These drugs are listed in Table 3.
| Drug Class | Drugs Within Class That Are Contraindicated With KALETRA | Clinical comments |
| Alpha 1- Adrenoreceptor antagonist | Alfuzosin | Potentially increased alfuzosin concentrations can result in hypotension. |
| Antimycobacterial | Rifampin | May lead to loss of virologic response and possible resistance to KALETRA or to the class of protease inhibitors or other co-administered antiretroviral agents [see Drug Interactions (7)]. |
| Ergot Derivatives | Dihydroergotamine, ergonovine, ergotamine, methylergonovine | Potential for acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. |
| GI motility agent | Cisapride | Potential for cardiac arrhythmias. |
| Herbal Products | St. John's Wort (hypericum perforatum) | May lead to loss of virologic response and possible resistance to KALETRA or to the class of protease inhibitors. |
| HMG-CoA Reductase Inhibitors | Lovastatin, simvastatin | Potential for myopathy including rhabdomyolysis. |
| PDE5 enzyme inhibitor | Sildenafila (Revatio®) when used for the treatment of pulmonary arterial hypertension | A safe and effective dose has not been established when used with KALETRA. There is an increased potential for sildenafil-associated adverse events, including visual abnormalities, hypotension, prolonged erection, and syncope [see Drug Interactions (7)] . |
| Neuroleptic | Pimozide | Potential for cardiac arrhythmias. |
| Sedative/Hypnotics |
Triazolam; orally administered midazolamb |
Prolonged or increased sedation or respiratory depression. |
WARNING AND PRECAUTIONS
KALETRA is a CYP3A inhibitor. Initiating treatment with KALETRA in patients receiving medications metabolized by CYP3A or initiating medications metabolized by CYP3A in patients already maintained on KALETRA may result in increased plasma concentrations of concomitant medications. Higher plasma concentrations of concomitant medications can result in increased or prolonged therapeutic or adverse effects, potentially leading to severe, life-threatening or fatal events. The potential for drug-drug interactions must be considered prior to and during therapy with KALETRA. Review of other medications taken by patients and monitoring of patients for adverse effects is recommended during therapy with KALETRA.
See Tables 3 and 9 for listing of drugs that are contraindicated for use with KALETRA due to potentially life-threatening adverse events, significant drug interactions, or loss of virologic activity [see Contraindications (4) and Drug Interactions (7)] .
Pancreatitis has been observed in patients receiving KALETRA therapy, including those who developed marked triglyceride elevations. In some cases, fatalities have been observed. Although a causal relationship to KALETRA has not been established, marked triglyceride elevations are a risk factor for development of pancreatitis [see Warnings and Precautions (5.9)] . Patients with advanced HIV-1 disease may be at increased risk of elevated triglycerides and pancreatitis, and patients with a history of pancreatitis may be at increased risk for recurrence during KALETRA therapy.
Pancreatitis should be considered if clinical symptoms (nausea, vomiting, abdominal pain) or abnormalities in laboratory values (such as increased serum lipase or amylase values) suggestive of pancreatitis occur. Patients who exhibit these signs or symptoms should be evaluated and KALETRA and/or other antiretroviral therapy should be suspended as clinically appropriate.
Patients with underlying hepatitis B or C or marked elevations in transaminase prior to treatment may be at increased risk for developing or worsening of transaminase elevations or hepatic decompensation with use of KALETRA.
There have been postmarketing reports of hepatic dysfunction, including some fatalities. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications in the setting of underlying chronic hepatitis or cirrhosis. A causal relationship with KALETRA therapy has not been established.
Elevated transaminases with or without elevated bilirubin levels have been reported in HIV-1 mono-infected and uninfected patients as early as 7 days after the initiation of KALETRA in conjunction with other antiretroviral agents. In some cases, the hepatic dysfunction was serious; however, a definitive causal relationship with KALETRA therapy has not been established.
Appropriate laboratory testing should be conducted prior to initiating therapy with KALETRA and patients should be monitored closely during treatment. Increased AST/ALT monitoring should be considered in the patients with underlying chronic hepatitis or cirrhosis, especially during the first several months of KALETRA treatment [see Use In Specific Populations (8.6)].
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during post-marketing surveillance in HIV-1 infected patients receiving protease inhibitor therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued protease inhibitor therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and a causal relationship between protease inhibitor therapy and these events has not been established.
Lopinavir/ritonavir prolongs the PR interval in some patients. Cases of second or third degree atrioventricular block have been reported. KALETRA should be used with caution in patients with underlying structural heart disease, pre-existing conduction system abnormalities, ischemic heart disease or cardiomyopathies, as these patients may be at increased risk for developing cardiac conduction abnormalities.
The impact on the PR interval of co-administration of KALETRA with other drugs that prolong the PR interval (including calcium channel blockers, beta-adrenergic blockers, digoxin and atazanavir) has not been evaluated. As a result, co-administration of KALETRA with these drugs should be undertaken with caution, particularly with those drugs metabolized by CYP3A. Clinical monitoring is recommended [see Clinical Pharmacology (12.3)] .
Postmarketing cases of QT interval prolongation and torsade de pointes have been reported although causality of KALETRA could not be established. Avoid use in patients with congenital long QT syndrome, those with hypokalemia, and with other drugs that prolong the QT interval [see Clinical Pharmacology (12.3)] .
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including KALETRA. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis) which may necessitate further evaluation and treatment.
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
Treatment with KALETRA has resulted in large increases in the concentration of total cholesterol and triglycerides [see Adverse Reactions (6.1)] . Triglyceride and cholesterol testing should be performed prior to initiating KALETRA therapy and at periodic intervals during therapy. Lipid disorders should be managed as clinically appropriate, taking into account any potential drug-drug interactions with KALETRA and HMG-CoA reductase inhibitors [see Contraindications (4) and Drug Interactions (7.3)].
Increased bleeding, including spontaneous skin hematomas and hemarthrosis have been reported in patients with hemophilia type A and B treated with protease inhibitors. In some patients additional factor VIII was given. In more than half of the reported cases, treatment with protease inhibitors was continued or reintroduced. A causal relationship between protease inhibitor therapy and these events has not been established.
Because the potential for HIV cross-resistance among protease inhibitors has not been fully explored in KALETRA-treated patients, it is unknown what effect therapy with KALETRA will have on the activity of subsequently administered protease inhibitors [see Clinical Pharmacology (12.4)].
KALETRA ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling.
- PR Interval Prolongation, QT Interval Prolongation [see Warnings and Precautions (5.5, 5.6)]
- Drug Interactions [see Warnings and Precautions (5.1)]
- Pancreatitis [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety profile of KALETRA in adults is primarily based on 1964 HIV-1 infected patients in clinical trials.
The most common adverse reaction was diarrhea, which was generally of mild to moderate severity. In study 730, the incidence of diarrhea of any severity during 48 weeks of therapy was 60% in patients receiving KALETRA tablets once daily compared to 57% in patients receiving KALETRA tablets twice daily. More patients receiving KALETRA tablets once daily (14, 4.2%) had ongoing diarrhea at the time of discontinuation as compared to patients receiving KALETRA tablets twice daily (6, 1.8%). In study 730, discontinuations due to any adverse reaction were 4.8% in patients receiving KALETRA tablets once daily as compared to 3% in patients receiving KALETRA tablets twice daily. In study 802, the incidence of diarrhea of any severity during 48 weeks of therapy was 50% in patients receiving KALETRA tablets once daily compared to 39% in patients receiving KALETRA tablets twice daily. Moderate or severe drug-related diarrhea occurred in 14% of patients receiving KALETRA tablets once daily as compared to 11% in patients receiving KALETRA tablets twice daily. At the time of discontinuation, 19 (6.3%) patients receiving KALETRA tablets once daily had ongoing diarrhea, as compared to 11 (3.7%) patients receiving KALETRA tablets twice daily. Discontinuations due to any adverse reaction occurred in 4.3% of patients receiving KALETRA tablets once daily compared to 7.0% in patients receiving KALETRA tablets twice daily. In study 863, discontinuations of randomized therapy due to adverse reactions were 3.4% in KALETRA-treated and 3.7% in nelfinavir-treated patients.
Treatment-emergent clinical adverse reactions of moderate or severe intensity in less then 2% of patients treated with combination therapy for up to 48 weeks (Studies 863 and 730) and for up to 360 weeks (Study 720) are presented in Table 4 (treatment-naïve patients); and for up to 48 weeks (Studies 888 and 802), 84 weeks (Study 957) and 144 weeks (Study 765) in Table 5 (protease inhibitor-experienced patients).
|
|
Study 863 (48 Weeks) |
Study 720 (360 Weeks) |
Study 730 (48 Weeks) |
||
|
|
KALETRA 400/100 mg Twice Daily + d4T + 3TC
(N = 326) |
Nelfinavir 750 mg Three Times Daily + d4T +
3TC (N = 327) |
KALETRA Twice Daily2 +
d4T + 3TC (N = 100) |
KALETRA 800/200 mg Once Daily + TDF +FTC (N=333) | KALETRA 400/100 mg Twice Daily + TDF +FTC (N=331) |
|
1 Includes adverse reactions of possible or probable relationship to study drug. 2 Includes adverse reaction data from dose group I (200/100 mg twice daily [N = 16] and 400/100 mg twice daily [N = 16]) and dose group II (400/100 mg twice daily [N = 35] and 400/200 mg twice daily [N = 33]). Within dosing groups, moderate to severe nausea of probable/possible relationship to KALETRA occurred at a higher rate in the 400/200 mg dose arm compared to the 400/100 mg dose arm in group II. Definitions: d4T = Stavudine; 3TC = Lamivudine; TDF = Tenofovir Disoproxil Fumarate; FTC = Emtricitabine |
|||||
| Endocrine Disorders |
|
|
|
|
|
| Hypogonadism | 0% | 0% | 2% | 0% | 0% |
| Gastrointestinal Disorders |
|
|
|
|
|
| Diarrhea | 16% | 17% | 28% | 17% | 15% |
| Nausea | 7% | 5% | 16% | 7% | 5% |
| Vomiting | 2% | 2% | 6% | 3% | 4% |
| Abdominal Pain | 4% | 3% | 11% | 1% | 1% |
| Dyspepsia | 2% | less then 1% | 6% | 0% | 0% |
| Flatulence | 2% | 1% | 4% | 1% | 1% |
| General Disorders and Administration Site Conditions |
|
|
|
|
|
| Asthenia | 4% | 3% | 9% | less then 1% | greater then 1% |
| Infections and Infestations |
|
|
|
|
|
| Bronchitis | 0% | 0% | 2% | 0% | greater then 1% |
| Investigations |
|
|
|
|
|
| Weight decreased | 1% | greater then 1% | 2% | 0% | greater then 1% |
| Metabolism and Nutrition Disorders |
|
|
|
|
|
| Anorexia | 1% | greater then 1% | 2% | greater then 1% | 1% |
| Musculoskeletal and Connective Tissue Disorders |
|
|
|
|
|
| Myalgia | 1% | 1% | 2% | 0% | 0% |
| Nervous System Disorders |
|
|
|
|
|
| Headache | 2% | 2% | 6% | 2% | 2% |
| Paresthesia | 1% | 1% | 2% | 0% | 0% |
| Psychiatric Disorders |
|
|
|
|
|
| Insomnia | 2% | 1% | 3% | 1% | 0% |
| Depression | 1% | 2% | 0% | 0% | 0% |
| Libido decreased | less then 1% | less then 1% | 2% | 0% | greater then 1% |
| Skin and Subcutaneous Tissue Disorders |
|
|
|
|
|
| Rash | 1% | 2% | 5% | greater then 1% | 1% |
| Vascular Disorders |
|
|
|
|
|
| Vasodilation | 0% | 0% | 3% | 0% | 0% |
|
|
Study 888 (48 Weeks) |
Study 9572 and
Study 7653
(84-144 Weeks) |
Study 802 (48 Weeks) |
||
|
|
KALETRA 400/100 mg Twice Daily + NVP + NRTIs
(N = 148) |
Investigator-selected protease inhibitor(s) +
NVP + NRTIs (N = 140) |
KALETRA Twice Daily + NNRTI + NRTIs (N = 127) |
KALETRA 800/200 mg Once Daily +NRTIs
(N=300) |
KALETRA 400/100 mg Twice Daily + NRTIs
(N=299) |
|
1 Includes adverse reactions of possible or probable relationship to study drug. 2 Includes adverse reaction data from patients receiving 400/100 mg twice daily (n = 29) or 533/133 mg twice daily (n = 28) for 84 weeks. Patients received KALETRA in combination with NRTIs and efavirenz. 3 Includes adverse reaction data from patients receiving 400/100 mg twice daily (n = 36) or 400/200 mg twice daily (n = 34) for 144 weeks. Patients received KALETRA in combination with NRTIs and nevirapine. Definitions: NVP = Nevirapine; NRTI = Nucleoside Reverse Transcriptase Inhibitors; NNRTI = Non-nucleoside Reverse Transcriptase Inhibitors |
|||||
| Gastrointestinal Disorders |
|
|
|
|
|
| Diarrhea | 7% | 9% | 23% | 14% | 11% |
| Nausea | 7% | 16% | 5% | 3% | 7% |
| Vomiting | 4% | 12% | 2% | 2% | 3% |
| Abdominal Pain | 2% | 2% | 4% | 2% | greater then 1% |
| Abdominal Pain Upper | N/A | N/A | N/A | 1% | 2% |
| Dyspepsia | 1% | 1% | 2% | 1% | greater then 1% |
| Flatulence | 1% | 2% | 2% | 1% | 1% |
| Dysphasia | 2% | 1% | 0% | 0% | 0% |
| General Disorders and Administration Site Conditions |
|
|
|
|
|
| Asthenia | 3% | 6% | 9% | less then 1% | greater then 1% |
| Pyrexia | 2% | 1% | 2% | 0% | less then 1% |
| Chills | 2% | 0% | 0% | 0% | 0% |
| Investigations |
|
|
|
|
|
| Weight decreased | 0% | 1% | 3% | greater then 1% | greater then 1% |
| Metabolism and Nutrition Disorders |
|
|
|
|
|
| Anorexia | 1% | 3% | 0% | 0% | 1% |
| Musculoskeletal and Connective Tissue Disorders |
|
|
|
|
|
| Myalgia | 1% | 1% | 2% | 0% | 0% |
| Nervous System Disorders |
|
|
|
|
|
| Headache | 2% | 3% | 2% | greater then 1% | 0% |
| Paresthesia | 0% | 1% | 2% | 0% | 0% |
| Psychiatric Disorders |
|
|
|
|
|
| Depression | 1% | 2% | 3% | greater then 1% | 0% |
| Insomnia | 0% | 2% | 2% | 0% | greater then 1% |
| Skin and Subcutaneous Tissue Disorders |
|
|
|
|
|
| Rash | 2% | 1% | 2% | 0% | 0% |
| Vascular Disorders |
|
|
|
|
|
| Hypertension | 0% | 0% | 2% | 0% | 0% |
Less Common Adverse Reactions
Treatment-emergent adverse reactions occurring in less than 2% of adult patients receiving KALETRA in the clinical trials supporting approval and of at least moderate intensity are listed below by system organ class.
Blood and Lymphatic System Disorders
Anemia, leukopenia, lymphadenopathy, neutropenia, and splenomegaly.
Cardiac Disorders
Angina pectoris, atrial fibrillation, atrioventricular block, myocardial infarction, palpitations, and tricuspid valve incompetence.
Ear and Labyrinth Disorders
Hyperacusis, tinnitus, and vertigo.
Endocrine Disorders
Cushing's syndrome and hypothyroidism.
Eye Disorders
Eye disorder and visual disturbance.
Gastrointestinal Disorders
Abdominal discomfort, abdominal distension, abdomen pain lower, constipation, duodenitis, dry mouth, enteritis, enterocolitis, enterocolitis hemorrhagic, eructation, esophagitis, fecal incontinence, gastric disorder, gastric ulcer, gastritis, gastroesophageal reflux disease, hemorrhoids, mouth ulceration, pancreatitis, periodontitis, rectal hemorrhage, stomach discomfort, and stomatitis.
General Disorders and Administration Site Conditions
Chest pain, cyst, drug interaction, edema, edema peripheral, face edema, fatigue, hypertrophy, and malaise.
Hepatobiliary Disorders
Cholangitis, cholecystitis, cytolytic hepatitis, hepatic steatosis, hepatitis, hepatomegaly, jaundice, and liver tenderness.
Immune System Disorders
Drug hypersensitivity, hypersensitivity, and immune reconstitution syndrome.
Infections and Infestations
Bacterial infection, bronchopneumonia, cellulitis, folliculitis, furuncle, gastroenteritis, influenza, otitis media, perineal abscess, pharyngitis, rhinitis, sialoadenitis, sinusitis, and viral infection.
Investigations
Drug level increased, glucose tolerance decreased, and weight increased.
Metabolism and Nutrition Disorders
Decreased appetite, dehydration, diabetes mellitus, hypovitaminosis, increased appetite, lactic acidosis, lipomatosis, and obesity.
Musculoskeletal and Connective Tissue Disorders
Arthralgia, arthropathy, back pain, muscular weakness, osteoarthritis, osteonecrosis, and pain in extremity.
Neoplasms Benign, Malignant and Unspecified (incl Cysts and Polyps)
Benign neoplasm of skin, lipoma, and neoplasm.
Nervous System Disorders
Ageusia, amnesia, ataxia, balance disorder, cerebral infarction, convulsion, dizziness, dysgeusia, dyskinesia, encephalopathy, extrapyramidal disorder, facial palsy, hypertonia, migraine, neuropathy, neuropathy peripheral, somnolence, and tremor.
Psychiatric Disorders
Abnormal dreams, affect lability, agitation, anxiety, apathy, confusional state, disorientation, mood swings, nervousness, and thinking abnormal.
Renal and Urinary Disorders
Hematuria, nephritis, nephrolithiasis, renal disorder, urine abnormality, and urine odor abnormal.
Reproductive System and Breast Disorders
Breast enlargement, ejaculation disorder, erectile dysfunction, gynecomastia, and menorrhagia.
Respiratory, Thoracic and Mediastinal Disorders
Asthma, cough, dyspnea, and pulmonary edema.
Skin and Subcutaneous Tissue Disorders
Acne, alopecia, dermatitis acneiform, dermatitis allergic, dermatitis exfoliative, dry skin, eczema, hyperhidrosis, idiopathic capillaritis, nail disorder, pruritis, rash generalized, rash maculo-papular, seborrhea, skin discoloration, skin hypertrophy, skin striae, skin ulcer, and swelling face.
Vascular Disorders
Deep vein thrombosis, orthostatic hypotension, thrombophlebitis, varicose vein, and vasculitis.
Laboratory Abnormalities
The percentages of adult patients treated with combination therapy with Grade 3-4 laboratory abnormalities are presented in Table 6 (treatment-naïve patients) and Table 7 (treatment-experienced patients).
|
|
|
Study 863 (48 Weeks) |
Study 720 (360 Weeks) |
Study 730 (48 Weeks) |
||
| Variable | Limit1 |
KALETRA 400/100 mg Twice Daily + d4T +3TC (N = 326) |
Nelfinavir 750 mg Three Times Daily + d4T + 3TC (N = 327) |
KALETRA Twice Daily + d4T + 3TC (N = 100) |
KALETRA Once Daily + TDF +FTC (N=333) |
KALETRA Twice Daily + TDF +FTC (N=331) |
|
1 ULN = upper limit of the normal range; N/A = Not Applicable. 2 Criterion for Study 730 was less then5x ULN (AST/ALT). |
||||||
| Chemistry | High |
|
|
|
|
|
| Glucose | less then 250 mg/dL | 2% | 2% | 4% | 0% | greater then 1% |
| Uric Acid | less then 12 mg/dL | 2% | 2% | 5% | greater then 1% | 1% |
|
SGOT/ AST2 |
less then 180 U/L | 2% | 4% | 10% | 1% | 2% |
|
SGPT/ ALT2 |
less then 215 U/L | 4% | 4% | 11% | 1% | 1% |
| GGT | less then 300 U/L | N/A | N/A | 10% | N/A | N/A |
|
Total Cholesterol |
less then 300 mg/dL | 9% | 5% | 27% | 4% | 3% |
| Triglycerides | less then 750 mg/dL | 9% | 1% | 29% | 3% | 6% |
| Amylase | less then 2 x ULN | 3% | 2% | 4% | N/A | N/A |
| Lipase | less then2 x ULN | N/A | N/A | N/A | 3% | 5% |
| Chemistry | Low |
|
|
|
|
|
| Calculated Creatinine Clearance | greater then 50 mL/min | N/A | N/A | N/A | 2% | 2% |
| Hematology | Low |
|
|
|
|
|
| Neutrophils | greater then 0.75 x 109/L | 1% | 3% | 5% | 2% | 1% |
|
|
|
Study 888 (48 Weeks) |
Study 9572 and Study
7653
(84-144 Weeks) |
Study 802 (48 Weeks) |
||
| Variable | Limit1 |
KALETRA 400/100 mg Twice Daily + NVP + NRTIs (N = 148) |
Investigator-selected protease inhibitor(s) +
NVP + NRTIs (N = 140) |
KALETRA Twice Daily + NNRTI + NRTIs (N = 127) |
KALETRA 800/200 mg Once Daily +NRTIs (N=300) |
KALETRA 400/100 mg Twice Daily +NRTIs (N=299) |
|
1 ULN = upper limit of the normal range; N/A = Not Applicable. 2 Includes clinical laboratory data from patients receiving 400/100 mg twice daily (n = 29) or 533/133 mg twice daily (n = 28) for 84 weeks. Patients received KALETRA in combination with NRTIs and efavirenz. 3 Includes clinical laboratory data from patients receiving 400/100 mg twice daily (n = 36) or 400/200 mg twice daily (n = 34) for 144 weeks. Patients received KALETRA in combination with NRTIs and nevirapine. 4 Criterion for Study 802 was less then 5x ULN (AST/ALT). |
||||||
| Chemistry | High |
|
|
|
|
|
| Glucose | less then 250 mg/dL | 1% | 2% | 5% | 2% | 2% |
| Total Bilirubin | less then 3.48 mg/dL | 1% | 3% | 1% | 1% | 1% |
| SGOT/AST4 | less then 180 U/L | 5% | 11% | 8% | 3% | 2% |
| SGPT/ALT4 | less then 215 U/L | 6% | 13% | 10% | 2% | 2% |
| GGT | less then 300 U/L | N/A | N/A | 29% | N/A | N/A |
|
Total Cholesterol |
less then 300 mg/dL | 20% | 21% | 39% | 6% | 7% |
| Triglycerides | less then 750 mg/dL | 25% | 21% | 36% | 5% | 6% |
| Amylase | less then 2 x ULN | 4% | 8% | 8% | 4% | 4% |
| Lipase | less then 2 x ULN | N/A | N/A | N/A | 4% | 1% |
|
Creatine Phosphokinase |
less then 4 x ULN | N/A | N/A | N/A | 4% | 5% |
| Chemistry | Low |
|
|
|
|
|
|
Calculated Creatinine Clearance |
greater then 50 mL/min | N/A | N/A | N/A | 3% | 3% |
|
Inorganic Phosphorus |
greater then 1.5 mg/dL | 1% | 0% | 2% | 1% | greater then 1% |
| Hematology | Low |
|
|
|
|
|
| Neutrophils | greater then 0.75 x 109/L | 1% | 2% | 4% | 3% | 4% |
| Hemoglobin | greater then 80 g/L | 1% | 1% | 1% | 1% | 2% |
KALETRA oral solution dosed up to 300/75 mg/m2 has been studied in 100 pediatric patients 6 months to 12 years of age. The adverse reaction profile seen during Study 940 was similar to that for adult patients.
Dysgeusia (22%), vomiting (21%), and diarrhea (12%) were the most common adverse reactions of any severity reported in pediatric patients treated with combination therapy for up to 48 weeks in Study 940. A total of 8 patients experienced adverse reactions of moderate to severe intensity. The adverse reactions meeting these criteria and reported for the 8 subjects include: hypersensitivity (characterized by fever, rash and jaundice), pyrexia, viral infection, constipation, hepatomegaly, pancreatitis, vomiting, alanine aminotransferase increased, dry skin, rash, and dysgeusia. Rash was the only event of those listed that occurred in 2 or more subjects (N = 3).
KALETRA oral solution dosed at 300/75 mg/m2 has been studied in 31 pediatric patients 14 days to 6 months of age. The adverse reaction profile in Study 1030 was similar to that observed in older children and adults. No adverse reaction was reported in greater than 10% of subjects. Adverse drug reactions of moderate to severe intensity occurring in 2 or more subjects included decreased neutrophil count (N=3), anemia (N=2), high potassium (N=2), and low sodium (N=2).
KALETRA oral solution and soft gelatin capsules dosed at higher than recommended doses including 400/100 mg/m2 (without concomitant NNRTI) and 480/120 mg/m2 (with concomitant NNRTI) have been studied in 26 pediatric patients 7 to 18 years of age in Study 1038. Patients also had saquinavir mesylate added to their regimen at Week 4. Rash (12%), blood cholesterol abnormal (12%) and blood triglycerides abnormal (12%) were the only adverse reactions reported in greater than 10% of subjects. Adverse drug reactions of moderate to severe intensity occurring in 2 or more subjects included rash (N=3), blood triglycerides abnormal (N=3), and electrocardiogram QT prolonged (N=2). Both subjects with QT prolongation had additional predisposing conditions such as electrolyte abnormalities, concomitant medications, or pre-existing cardiac abnormalities.
Laboratory Abnormalities
The percentages of pediatric patients treated with combination therapy including KALETRA with Grade 3-4 laboratory abnormalities are presented in Table 8.
| Variable | Limit1 |
KALETRA Twice Daily + RTIs (N = 100) |
|
1 ULN = upper limit of the normal range. 2 Subjects with Grade 3-4 amylase confirmed by elevations in pancreatic amylase. |
||
| Chemistry | High |
|
| Sodium | less then 149 mEq/L | 3% |
| Total Bilirubin | less then 3.0 x ULN | 3% |
| SGOT/AST | less then 180 U/L | 8% |
| SGPT/ALT | less then 215 U/L | 7% |
| Total Cholesterol | less then 300 mg/dL | 3% |
| Amylase | less then 2.5 x ULN | 7%2 |
| Chemistry | Low |
|
| Sodium | greater then 130 mEq/L | 3% |
| Hematology | Low |
|
| Platelet Count | greater then 50 x 109/L | 4% |
| Neutrophils | greater then 0.40 x 109/L | 2% |
The following adverse reactions have been reported during postmarketing use of KALETRA. Because these reactions are reported voluntarily from a population of unknown size, it is not possible to reliably estimate their frequency or establish a causal relationship to KALETRA exposure.
Body as a Whole
Redistribution/accumulation of
body fat has been reported
[see Warnings and Precautions (5.8)]
.
Cardiovascular
Bradyarrhythmias. First–degree
AV block, second-degree AV block, third-degree AV block, QTc interval
prolongation, torsades (torsade) de pointes
[see Warnings
and Precautions (5.5,5.6)]
.
Skin and Appendages
Toxic epidermal necrolysis
(TEN), Stevens Johnson Syndrome and erythema multiforme.
DRUG INTERACTIONS
See also Contraindications (4), Warnings and Precautions (5.1), Clinical Pharmacology (12.3)
Lopinavir/ritonavir is an inhibitor of CYP3A and may increase plasma concentrations of agents that are primarily metabolized by CYP3A. Agents that are extensively metabolized by CYP3A and have high first pass metabolism appear to be the most susceptible to large increases in AUC (less then 3-fold) when co-administered with KALETRA. Thus, co-administration of KALETRA with drugs highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events is contraindicated. Co-administration with other CYP3A substrates may require a dose adjustment or additional monitoring as shown in Table 9.
Additionally, KALETRA induces glucuronidation.
Lopinavir/ritonavir is a CYP3A substrate; therefore, drugs that induce CYP3A may decrease lopinavir plasma concentrations and reduce KALETRA’s therapeutic effect. Although not observed in the KALETRA/ketoconazole drug interaction study, co-administration of KALETRA and other drugs that inhibit CYP3A may increase lopinavir plasma concentrations.
Table 9 provides a listing of established or potentially clinically significant drug interactions. Alteration in dose or regimen may be recommended based on drug interaction studies or predicted interaction [see Clinical Pharmacology (12.3) for magnitude of interaction] .
|
Concomitant Drug Class:
Drug Name |
Effect on Concentration of Lopinavir or Concomitant Drug | Clinical Comment |
|
* see Clinical Pharmacology (12.3) for Magnitude of Interaction. |
||
| HIV-1 Antiviral Agents | ||
|
Non-nucleoside Reverse Transcriptase
Inhibitors: efavirenz*, nevirapine* |
↓ lopinavir |
KALETRA dose increase is recommended in all patients
[see Dosage and Administration (2.1)
and Clinical Pharmacology (12.3)]
. Increasing the dose of KALETRA tablets to 500/125 mg (given as two 200/50 mg tablets and one 100/25 mg tablet) twice daily co-administered with efavirenz resulted in similar lopinavir concentrations compared to KALETRA tablets 400/100 mg (given as two 200/50 mg tablets) twice daily without efavirenz. Increasing the dose of KALETRA tablets to 600/150 mg (given as three 200/50 mg tablets) twice daily co-administered with efavirenz resulted in significantly higher lopinavir plasma concentrations compared to KALETRA tablets 400/100 mg twice daily without efavirenz. KALETRA should not be administered once daily in combination with efavirenz or nevirapine [see Dosage and Administration (2.1) and Clinical Pharmacology (12.3)] . |
|
Non-nucleoside Reverse Transcriptase Inhibitor: delavirdine |
↑ lopinavir | Appropriate doses of the combination with respect to safety and efficacy have not been established. |
|
Nucleoside Reverse Transcriptase Inhibitor: didanosine |
|
KALETRA tablets can be administered simultaneously with didanosine without
food. For KALETRA oral solution, it is recommended that didanosine be administered on an empty stomach; therefore, didanosine should be given one hour before or two hours after KALETRA oral solution (given with food). |
|
Nucleoside Reverse Transcriptase Inhibitor: tenofovir |
↑ tenofovir | KALETRA increases tenofovir concentrations. The mechanism of this interaction is unknown. Patients receiving KALETRA and tenofovir should be monitored for adverse reactions associated with tenofovir. |
|
Nucleoside Reverse Transcriptase Inhibitor: abacavir zidovudine |
↓ abacavir ↓ zidovudine |
KALETRA induces glucuronidation; therefore, KALETRA has the potential to reduce zidovudine and abacavir plasma concentrations. The clinical significance of this potential interaction is unknown. |
|
HIV-1 Protease Inhibitor: amprenavir* |
↑ amprenavir ↓ lopinavir |
KALETRA should not be administered once daily in combination with amprenavir
[see Dosage and Administration (2.1)] . |
|
HIV-1 Protease Inhibitor: fosamprenavir/ritonavir |
↓ amprenavir ↓ lopinavir |
An increased rate of adverse reactions has been observed with co-administration of these medications. Appropriate doses of the combinations with respect to safety and efficacy have not been established. |
|
HIV-1 Protease Inhibitor: indinavir* |
↑ indinavir | Decrease indinavir dose to 600 mg twice daily, when co-administered with KALETRA 400/100 mg twice daily [see Clinical Pharmacology (12.3)] . KALETRA once daily has not been studied in combination with indinavir. |
|
HIV-1 Protease Inhibitor: nelfinavir* |
↑ nelfinavir ↑ M8 metabolite of nelfinavir ↓ lopinavir |
KALETRA should not be administered once daily in combination with
nelfinavir [see Dosage and Administration (2.1) and Clinical Pharmacology (12.3)] . |
|
HIV-1 Protease Inhibitor: ritonavir* |
↑ lopinavir | Appropriate doses of additional ritonavir in combination with KALETRA with respect to safety and efficacy have not been established. |
|
HIV-1 Protease Inhibitor: saquinavir* |
↑ saquinavir |
The saquinavir dose is 1000 mg twice daily, when co-administered with
KALETRA 400/100 mg twice daily. KALETRA once daily has not been studied in combination with saquinavir. |
|
HIV-1 Protease Inhibitor: tipranavir |
↓ lopinavir AUC and Cmin | KALETRA should not be administered with tipranavir (500 mg twice daily) co-administered with ritonavir (200 mg twice daily). |
| HIV CCR5 – antagonist: maraviroc | ↑ maraviroc | Concurrent administration of maraviroc with KALETRA will increase plasma levels of maraviroc. When co-administered, patients should receive 150 mg twice daily of maraviroc. For further details see complete prescribing information for Selzentry® (maraviroc). |
| Other Agents | ||
|
Antiarrhythmics: amiodarone, bepridil, lidocaine (systemic), and quinidine |
↑ antiarrhythmics | Caution is warranted and therapeutic concentration monitoring (if available) is recommended for antiarrhythmics when co-administered with KALETRA. |
|
Anticancer Agents: vincristine, vinblastine, dasatinib, nilotinib |
↑ anticancer agents |
Concentrations of these drugs may be increased when co-administered with
KALETRA resulting in the potential for increased adverse events usually
associated with these anticancer agents. For vincristine and vinblastine, consideration should be given to temporarily withholding the ritonavir-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when KALETRA is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consideration should be given to initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. A decrease in the dosage or an adjustment of the dosing interval of nilotinib and dasatinib may be necessary for patients requiring co-administration with strong CYP3A inhibitors such as KALETRA. Please refer to the nilotinib and dasatinib prescribing information for dosing instructions. |
|
Anticoagulant: warfarin |
|
Concentrations of warfarin may be affected. It is recommended that INR (international normalized ratio) be monitored. |
|
Anticonvulsants: carbamazepine, phenobarbital, phenytoin |
↓ lopinavir ↓ phenytoin |
KALETRA may be less effective due to decreased lopinavir plasma
concentrations in patients taking these agents concomitantly and should be used
with caution. KALETRA should not be administered once daily in combination with carbamazepine, phenobarbital, or phenytoin. In addition, co-administration of phenytoin and KALETRA may cause decreases in steady-state phenytoin concentrations. Phenytoin levels should be monitored when co-administering with KALETRA. |
|
Antidepressant: bupropion |
↓ bupropion ↓ active metabolite, hydroxybupropion |
Concurrent administration of bupropion with KALETRA may decrease plasma levels of both bupropion and its active metabolite (hydroxybupropion). Patients receiving KALETRA and bupropion concurrently should be monitored for an adequate clinical response to bupropion. |
|
Antidepressant: trazodone |
↑ trazodone | Concomitant use of trazodone and KALETRA may increase concentrations of trazodone. Adverse reactions of nausea, dizziness, hypotension and syncope have been observed following co-administration of trazodone and ritonavir. If trazodone is used with a CYP3A4 inhibitor such as ritonavir, the combination should be used with caution and a lower dose of trazodone should be considered. |
|
Anti-infective: clarithromycin |
↑ clarithromycin |
For patients with renal impairment, the following dosage adjustments should
be considered:
No dose adjustment for patients with normal renal function is necessary. |
|
Antifungals: ketoconazole*, itraconazole, voriconazole |
↑ ketoconazole ↑ itraconazole ↓ voriconazole |
High doses of ketoconazole (less then 200 mg/day) or itraconazole
(less then 200 mg/day) are not recommended. Co-administration of voriconazole with KALETRA has not been studied. However, a study has been shown that administration of voriconazole with ritonavir 100 mg every 12 hours decreased voriconazole steady-state AUC by an average of 39%; therefore, co-administration of KALETRA and voriconazole may result in decreased voriconazole concentrations and the potential for decreased voriconazole effectiveness and should be avoided, unless an assessment of the benefit/risk to the patient justifies the use of voriconazole. Otherwise, alternative antifungal therapies should be considered in these patients. |
|
Anti-gout: colchicine |
↑ colchicine |
Patients with renal or hepatic impairment should not be given colchicine
with KALETRA.
Treatment of gout flares-co-administration of colchicine in patients on KALETRA: 0.6 mg (1 tablet) x 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Dose to be repeated no earlier than 3 days. Prophylaxis of gout flares-co-administration of colchicine in patients on KALETRA: If the original colchicine regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day. If the original colchicine regimen was 0.6 mg once a day, the regimen should be adjusted to 0.3 mg once every other day. Treatment of familial Mediterranean fever (FMF)-co-administration of colchicine in patients on KALETRA: Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day). |
|
Antimycobacterial: rifabutin* |
↑ rifabutin and rifabutin metabolite | Dosage reduction of rifabutin by at least 75% of the usual dose of 300 mg/day is recommended (i.e., a maximum dose of 150 mg every other day or three times per week). Increased monitoring for adverse reactions is warranted in patients receiving the combination. Further dosage reduction of rifabutin may be necessary. |
|
Antimycobacterial: rifampin |
↓ lopinavir | May lead to loss of virologic response and possible resistance to KALETRA or to the class of protease inhibitors or other co-administered antiretroviral agents. A study evaluated combination of rifampin 600 mg once daily, with KALETRA 800/200 mg twice daily or KALETRA 400/100 mg + ritonavir 300 mg twice daily. Pharmacokinetic and safety results from this study do not allow for a dose recommendation. Nine subjects (28%) experienced a ≥ grade 2 increase in ALT/AST, of which seven (21%) prematurely discontinued study per protocol. Based on the study design, it is not possible to determine whether the frequency or magnitude of the ALT/AST elevations observed is higher than what would be seen with rifampin alone [see Clinical Pharmacology (12.3) for magnitude of interaction] . |
|
Antiparasitic: atovaquone |
↓ atovaquone | Clinical significance is unknown; however, increase in atovaquone doses may be needed. |
| Benzodiazepines: parenterally administered midazolam | ↑ midazolam | Midazolam is extensively metabolized by CYP3A4. Increases in the concentration of midazolam are expected to be significantly higher with oral than parenteral administration. Therefore, KALETRA should not be given with orally administered midazolam [see Contraindications (4)] . If KALETRA is coadministered with parenteral midazolam, close clinical monitoring for respiratory depression and/or prolonged sedation should be exercised and dosage adjustment should be considered. |
|
Calcium Channel Blockers, dihydropyridine: e.g., felodipine, nifedipine, nicardipine |
↑ dihydropyridine calcium channel blockers | Caution is warranted and clinical monitoring of patients is recommended. |
|
Contraceptive: ethinyl estradiol* |
↓ ethinyl estradiol | Because contraceptive steroid concentrations may be altered when KALETRA is co-administered with oral contraceptives or with the contraceptive patch, alternative methods of nonhormonal contraception are recommended. |
|
Corticosteroid: dexamethasone |
↓ lopinavir |
Use with caution. KALETRA may be less effective due to decreased lopinavir
plasma concentrations in patients taking these agents
concomitantly. |
| disulfiram/metronidazole |
|
KALETRA oral solution contains alcohol, which can produce disulfiram-like reactions when co-administered with disulfiram or other drugs that produce this reaction (e.g., metronidazole). |
|
Endothelin receptor antagonists: bosentan |
↑ bosentan |
Co-administration of bosentan in patients on
KALETRA:
In patients who have been receiving KALETRA for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Co-administration of KALETRA in patients on bosentan: Discontinue use of bosentan at least 36 hours prior to initiation of KALETRA. After at least 10 days following the initiation of KALETRA, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability. |
|
HMG-CoA Reductase Inhibitors: atorvastatin rosuvastatin |
↑ atorvastatin ↑ rosuvastatin |
Use lowest possible dose of atorvastatin or rosuvastatin with careful monitoring, or consider other HMG-CoA reductase inhibitors such as pravastatin or fluvastatin in combination with KALETRA. |
|
Immunosuppressants: cyclosporine, tacrolimus, rapamycin |
↑ immunosuppressants | Therapeutic concentration monitoring is recommended for immunosuppressant agents when co-administered with KALETRA. |
|
Inhaled Steroid: fluticasone |
↑ fluticasone | Concomitant use of fluticasone propionate and KALETRA may increase plasma concentrations of fluticasone propionate, resulting in significantly reduced serum cortisol concentrations. Systemic corticosteroid effects including Cushing's syndrome and adrenal suppression have been reported during post-marketing use in patients receiving ritonavir and inhaled or intranasally administered fluticasone propionate. Co-administration of fluticasone propionate and KALETRA is not recommended unless the potential benefit to the patient outweighs the risk of systemic corticosteroid side effect. |
|
Long-acting beta-adrenoceptor agonist: salmeterol |
↑ salmeterol | Concurrent administration of salmeterol and KALETRA is not recommended. The combination may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations and sinus tachycardia. |
|
Narcotic Analgesic: methadone* fentanyl |
↓ methadone ↑ fentanyl |
Dosage of methadone may need to be increased when co-administered with
KALETRA. Concentrations of fentanyl are expected to increase. Careful monitoring of therapeutic and adverse effects (including potentially fatal respiratory depression) is recommended when fentanyl is concomitantly administered with KALETRA. |
|
PDE5 inhibitors: sildenafil, tadalafil, vardenafil |
↑ sildenafil ↑ tadalafil ↑ vardenafil |
Particular caution should be used when prescribing sildenafil, tadalafil, or
vardenafil in patients receiving KALETRA. Co-administration of KALETRA with
these drugs is expected to substantially increase their concentrations and may
result in an increase in PDE5 inhibitor associated adverse reactions including
hypotension, syncope, visual changes and prolonged erection.
Use of PDE5 inhibitors for pulmonary arterial hypertension (PAH): Sildenafil (Revatio®) is contraindicated when used for the treatment of pulmonary arterial hypertension (PAH) because a safe and effective dose has not been established when used with KALETRA [see Contraindications (4)] . The following dose adjustments are recommended for use of tadalafil (Adcirca®) with KALETRA: Co-administration of ADCIRCA in patients on KALETRA: In patients receiving KALETRA for at least one week, start ADCIRCA at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Co-administration of KALETRA in patients on ADCIRCA: Avoid use of ADCIRCA during the initiation of KALETRA. Stop ADCIRCA at least 24 hours prior to starting KALETRA. After at least one week following the initiation of KALETRA, resume ADCIRCA at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Use of PDE5 inhibitors for erectile dysfunction: It is recommended not to exceed the following doses:
|
Drug interaction studies reveal no clinically significant interaction between KALETRA and desipramine (CYP2D6 probe), pravastatin, stavudine, lamivudine, omeprazole or ranitidine.
Based on known metabolic profiles, clinically significant drug interactions are not expected between KALETRA and fluvastatin, dapsone, trimethoprim/sulfamethoxazole, azithromycin, erythromycin, or fluconazole.
USE IN SPECIFIC POPULATIONS
Pregnancy Category C.
No treatment-related malformations were observed when lopinavir in combination with ritonavir was administered to pregnant rats or rabbits. Embryonic and fetal developmental toxicities (early resorption, decreased fetal viability, decreased fetal body weight, increased incidence of skeletal variations and skeletal ossification delays) occurred in rats at a maternally toxic dosage. Based on AUC measurements, the drug exposures in rats at the toxic doses were approximately 0.7-fold for lopinavir and 1.8-fold for ritonavir for males and females that of the exposures in humans at the recommended therapeutic dose (400/100 mg twice daily). In a peri- and postnatal study in rats, a developmental toxicity (a decrease in survival in pups between birth and postnatal Day 21) occurred.
No embryonic and fetal developmental toxicities were observed in rabbits at a maternally toxic dosage. Based on AUC measurements, the drug exposures in rabbits at the toxic doses were approximately 0.6-fold for lopinavir and 1.0-fold for ritonavir that of the exposures in humans at the recommended therapeutic dose (400/100 mg twice daily). There are, however, no adequate and well-controlled studies in pregnant women. KALETRA should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Antiretroviral Pregnancy Registry : To monitor maternal-fetal outcomes of pregnant women exposed to KALETRA, an Antiretroviral Pregnancy Registry has been established. Physicians are encouraged to register patients by calling 1-800-258-4263.
T he Centers for Disease Control and Prevention recommend that HIV-1 infected mothers not breast-feed their infants to avoid risking postnatal transmission of HIV-1. Studies in rats have demonstrated that lopinavir is secreted in milk. It is not known whether lopinavir is secreted in human milk. Because of both the potential for HIV-1 transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breast-feed if they are receiving KALETRA.
The safety, efficacy, and pharmacokinetic profiles of KALETRA in pediatric patients below the age of 14 days have not been established. KALETRA once daily has not been evaluated in pediatric patients.
An open-label, multi-center, dose-finding trial was performed to evaluate the pharmacokinetic profile, tolerability, safety and efficacy of KALETRA oral solution containing lopinavir 80 mg/mL and ritonavir 20 mg/mL at a dose of with 300/75 mg/m2 twice daily plus two NRTIs in HIV-infected infants less then14 days and greater then 6 months of age. Results revealed that infants younger than 6 months of age generally had lower lopinavir AUC12 than older children (6 months to 12 years of age), however, despite the lower lopinavir drug exposure observed, antiviral activity was demonstrated as reflected in the proportion of subjects who achieved HIV-RNA greater then 400 copies/mL at Week 24 [see Adverse Reactions,Clinical Pharmacology, Clinical Studies].
Safety and efficacy in pediatric patients less then 6 months of age was demonstrated in a clinical trial in 100 patients. The clinical trial was an open-label, multicenter trial evaluating the pharmacokinetic profile, tolerability, safety, and efficacy of KALETRA oral solution containing lopinavir 80 mg/mL and ritonavir 20 mg/mL in 100 antiretroviral naïve and experienced pediatric patients ages 6 months to 12 years. Dose selection for patients 6 months to 12 years of age was based on the following results. The 230/57.5 mg/m2 oral solution twice daily regimen without nevirapine and the 300/75 mg/m2 oral solution twice daily regimen with nevirapine provided lopinavir plasma concentrations similar to those obtained in adult patients receiving the 400/100 mg twice daily regimen (without nevirapine) [see Adverse Reactions), Clinical Pharmacology), Clinical Studies].
A prospective multicenter, open-label trial evaluated the pharmacokinetic profile, tolerability, safety and efficacy of high-dose KALETRA with or without concurrent NNRTI therapy (Group 1: 400/100 mg/m2 twice daily + less then 2 NRTIs; Group 2: 480/120 mg/m2 twice daily + less then 1 NRTI + 1 NNRTI) in children and adolescents less then 2 years to greater then 18 years of age who had failed prior therapy. Patients also had saquinavir mesylate added to their regimen. This strategy was intended to assess whether higher than approved doses of KALETRA could overcome protease inhibitor cross-resistance. High doses of KALETRA exhibited a safety profile similar to those observed in previous trials; changes in HIV-1 RNA were less than anticipated; three patients had HIV-1 RNA greater then 400 copies/mL at Week 48. CD4+ cell count increases were noted in the eight patients who remained on treatment for 48 weeks [see Adverse Reactions), Clinical Pharmacology] .
Clinical studies of KALETRA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, appropriate caution should be exercised in the administration and monitoring of KALETRA in elderly patients reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
KALETRA is principally metabolized by the liver; therefore, caution should be exercised when administering this drug to patients with hepatic impairment, because lopinavir concentrations may be increased [see Warnings and Precautions) and Clinical Pharmacology] .
OVERDOSAGE
Overdoses with KALETRA oral solution have been reported. One of these reports described fatal cardiogenic shock in a 2.1 kg infant who received a single dose of 6.5 mL of KALETRA oral solution nine days prior. However, a causal relationship between the overdose and the outcome could not be established. Healthcare professionals should be aware that KALETRA oral solution is highly concentrated and therefore, should pay special attention to accurate calculation of the dose of KALETRA, transcription of the medication order, dispensing information and dosing instructions to minimize the risk for medication errors and overdose. This is especially important for infants and young children.
KALETRA oral solution contains 42.4% alcohol (v/v). Accidental ingestion of the product by a young child could result in significant alcohol-related toxicity and could approach the potential lethal dose of alcohol.
Human experience of acute overdosage with KALETRA is limited. Treatment of overdose with KALETRA should consist of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. There is no specific antidote for overdose with KALETRA. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage. Administration of activated charcoal may also be used to aid in removal of unabsorbed drug. Since KALETRA is highly protein bound, dialysis is unlikely to be beneficial in significant removal of the drug.
KALETRA DESCRIPTION
KALETRA (lopinavir/ritonavir) is a co-formulation of lopinavir and ritonavir. Lopinavir is an inhibitor of the HIV-1 protease. As co-formulated in KALETRA, ritonavir inhibits the CYP3A-mediated metabolism of lopinavir, thereby providing increased plasma levels of lopinavir.
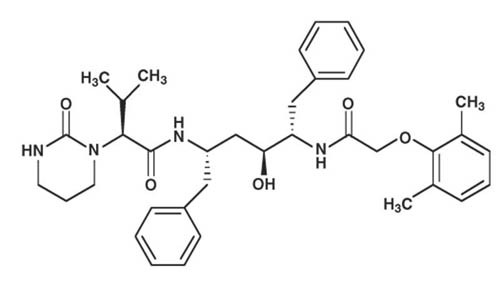
Lopinavir is chemically designated as [1S-[1R*,(R*), 3R*, 4R*]]-N-[4-[[(2,6-dimethylphenoxy)acetyl]amino]-3-hydroxy-5-phenyl-1-(phenylmethyl)pentyl]tetrahydro-alpha-(1-methylethyl)-2-oxo-1(2H)-pyrimidineacetamide. Its molecular formula is C37H48N4O5, and its molecular weight is 628.80. Lopinavir is a white to light tan powder. It is freely soluble in methanol and ethanol, soluble in isopropanol and practically insoluble in water. Lopinavir has the following structural formula:
- Yellow tablets containing 200 mg of lopinavir and 50 mg of ritonavir
- Pale yellow tablets containing 100 mg of lopinavir and 25 mg of ritonavir.
The yellow, 200 mg lopinavir/50 mg ritonavir, tablets contain the following inactive ingredients: copovidone, sorbitan monolaurate, colloidal silicon dioxide, and sodium stearyl fumarate. The following are the ingredients in the film coating: hypromellose, titanium dioxide, polyethylene glycol 400, hydroxypropyl cellulose, talc, colloidal silicon dioxide, polyethylene glycol 3350, yellow ferric oxide E172, and polysorbate 80.
The pale yellow, 100 mg lopinavir/25 mg ritonavir, tablets contain the following inactive ingredients: copovidone, sorbitan monolaurate, colloidal silicon dioxide, and sodium stearyl fumarate. The following are the ingredients in the film coating: polyvinyl alcohol, titanium dioxide, talc, polyethylene glycol 3350, and yellow ferric oxide E172.
KALETRA oral solution is available for oral administration as 80 mg lopinavir and 20 mg ritonavir per milliliter with the following inactive ingredients: acesulfame potassium, alcohol, artificial cotton candy flavor, citric acid, glycerin, high fructose corn syrup, Magnasweet-110 flavor, menthol, natural and artificial vanilla flavor, peppermint oil, polyoxyl 40 hydrogenated castor oil, povidone, propylene glycol, saccharin sodium, sodium chloride, sodium citrate, and water.
KALETRA oral solution contains 42.4% alcohol (v/v).
CLINICAL PHARMACOLOGY
Lopinavir is an antiviral drug [see Clinical Pharmacology] .
The pharmacokinetic properties of lopinavir co-administered with ritonavir have been evaluated in healthy adult volunteers and in HIV-1 infected patients; no substantial differences were observed between the two groups. Lopinavir is essentially completely metabolized by CYP3A. Ritonavir inhibits the metabolism of lopinavir, thereby increasing the plasma levels of lopinavir. Across studies, administration of KALETRA 400/100 mg twice daily yields mean steady-state lopinavir plasma concentrations 15- to 20-fold higher than those of ritonavir in HIV-1 infected patients. The plasma levels of ritonavir are less than 7% of those obtained after the ritonavir dose of 600 mg twice daily. The in vitro antiviral EC50 of lopinavir is approximately 10-fold lower than that of ritonavir. Therefore, the antiviral activity of KALETRA is due to lopinavir.
Absorption
In a pharmacokinetic study in HIV-1
positive subjects (n = 19), multiple dosing with 400/100 mg KALETRA twice daily
with food for 3 weeks produced a mean SD lopinavir peak plasma concentration
(Cmax) of 9.8 ± 3.7 µg/mL, occurring approximately 4
hours after administration. The mean steady-state trough concentration prior to
the morning dose was 7.1 ± 2.9 µg/mL and minimum concentration within a dosing
interval was 5.5 ± 2.7 µg/mL. Lopinavir AUC over a 12 hour dosing interval
averaged 92.6 ± 36.7 µg•h/mL. The absolute
bioavailability of lopinavir co-formulated with ritonavir in humans has not been
established. Under nonfasting conditions (500 kcal, 25% from fat), lopinavir
concentrations were similar following administration of KALETRA co-formulated
capsules and oral solution. When administered under fasting conditions, both the
mean AUC and Cmax of lopinavir were 22% lower for the
KALETRA oral solution relative to the capsule formulation.
Plasma concentrations of lopinavir and ritonavir after administration of two 200/50 mg KALETRA tablets are similar to three 133.3/33.3 mg KALETRA capsules under fed conditions with less pharmacokinetic variability.
Effects of Food on Oral Absorption
KALETRA Tablets
No clinically significant changes in
Cmax and AUC were observed following administration of
KALETRA tablets under fed conditions compared to fasted conditions. Relative to
fasting, administration of KALETRA tablets with a moderate fat meal (500 - 682
Kcal, 23 to 25% calories from fat) increased lopinavir AUC and Cmax by 26.9% and 17.6%, respectively. Relative to fasting,
administration of KALETRA tablets with a high fat meal (872 Kcal, 56% from fat)
increased lopinavir AUC by 18.9% but not Cmax. Therefore,
KALETRA tablets may be taken with or without food.
KALETRA Oral Solution
Relative to fasting,
administration of KALETRA oral solution with a moderate fat meal (500 - 682
Kcal, 23 to 25% calories from fat) increased lopinavir AUC and Cmax by 80 and 54%, respectively. Relative to fasting,
administration of KALETRA oral solution with a high fat meal (872 Kcal, 56% from
fat) increased lopinavir AUC and Cmax by 130% and 56%,
respectively. To enhance bioavailability and minimize pharmacokinetic
variability KALETRA oral solution should be taken with food.
Distribution
At steady state, lopinavir is
approximately 98-99% bound to plasma proteins. Lopinavir binds to both
alpha-1-acid glycoprotein (AAG) and albumin; however, it has a higher affinity
for AAG. At steady state, lopinavir protein binding remains constant over the
range of observed concentrations after 400/100 mg KALETRA twice daily, and is
similar between healthy volunteers and HIV-1 positive patients.
Metabolism
In vitro
experiments with human hepatic microsomes indicate that lopinavir primarily
undergoes oxidative metabolism. Lopinavir is extensively metabolized by the
hepatic cytochrome P450 system, almost exclusively by the CYP3A isozyme.
Ritonavir is a potent CYP3A inhibitor which inhibits the metabolism of
lopinavir, and therefore increases plasma levels of lopinavir. A 14C-lopinavir study in humans showed that 89% of the plasma
radioactivity after a single 400/100 mg KALETRA dose was due to parent drug. At
least 13 lopinavir oxidative metabolites have been identified in man. Ritonavir
has been shown to induce metabolic enzymes, resulting in the induction of its
own metabolism. Pre-dose lopinavir concentrations decline with time during
multiple dosing, stabilizing after approximately 10 to 16 days.
Elimination
Following a 400/100 mg 14C-lopinavir/ritonavir dose, approximately 10.4 ± 2.3% and
82.6 ± 2.5% of an administered dose of 14C-lopinavir can
be accounted for in urine and feces, respectively, after 8 days. Unchanged
lopinavir accounted for approximately 2.2 and 19.8% of the administered dose in
urine and feces, respectively. After multiple dosing, less than 3% of the
lopinavir dose is excreted unchanged in the urine. The apparent oral clearance
(CL/F) of lopinavir is 5.98 ± 5.75 L/hr (mean ± SD, n = 19).
Once Daily Dosing
The pharmacokinetics of once
daily KALETRA have been evaluated in HIV-1 infected subjects naïve to
antiretroviral treatment. KALETRA 800/200 mg was administered in combination
with emtricitabine 200 mg and tenofovir DF 300 mg as part of a once daily
regimen. Multiple dosing of 800/200 mg KALETRA once daily for 4 weeks with food
(n = 24) produced a mean ± SD lopinavir peak plasma concentration (Cmax) of 11.8 ± 3.7 µg/mL, occurring approximately 6 hours after
administration. The mean steady-state lopinavir trough concentration prior to
the morning dose was 3.2 ± 2.1 µg/mL and minimum concentration within a dosing
interval was 1.7 ± 1.6 µg/mL. Lopinavir AUC over a 24 hour dosing interval
averaged 154.1 ± 61.4 µg• h/mL.
The pharmacokinetics of once daily KALETRA has also been evaluated in treatment experienced HIV-1 infected subjects. Lopinavir exposure (Cmax, AUC[0-24h], Ctrough) with once daily KALETRA administration in treatment experienced subjects is comparable to the once daily lopinavir exposure in treatment naïve subjects.
Effects on Electrocardiogram
QTcF interval was evaluated in a randomized, placebo and active (moxifloxacin 400 mg once daily) controlled crossover study in 39 healthy adults, with 10 measurements over 12 hours on Day 3. The maximum mean time-matched (95% upper confidence bound) differences in QTcF interval from placebo after baseline-correction were 5.3 (8.1) and 15.2 (18.0) mseconds (msec) for 400/100 mg twice daily and supratherapeutic 800/200 mg twice daily KALETRA, respectively. KALETRA 800/200 mg twice daily resulted in a Day 3 mean Cmax approximately 2-fold higher than the mean Cmax observed with the approved once daily and twice daily KALETRA doses at steady state.
PR interval prolongation was also noted in subjects receiving KALETRA in the same study on Day 3. The maximum mean (95% upper confidence bound) difference from placebo in the PR interval after baseline-correction were 24.9 (21.5, 28.3) and 31.9 (28.5, 35.3) msec for 400/100 mg twice daily and supratherapeutic 800/200 mg twice daily KALETRA, respectively [see Warnings and Precautions] .
Special Populations
Gender, Race and Age
No gender related
pharmacokinetic differences have been observed in adult patients. No clinically
important pharmacokinetic differences due to race have been identified.
Lopinavir pharmacokinetics have not been studied in elderly patients.
Pediatric Patients
The pharmacokinetics of
KALETRA oral solution 300/75 mg/m2 twice daily and
230/57.5 mg/m2 twice daily have been studied in a total
of 53 pediatric patients in Study 940, ranging in age from 6 months to 12 years
[see Clinical Studies]. The 230/57.5 mg/m2 twice daily regimen without nevirapine and the 300/75
mg/m2 twice daily regimen with nevirapine provided
lopinavir plasma concentrations similar to those obtained in adult patients
receiving the 400/100 mg twice daily regimen (without nevirapine).
The mean steady-state lopinavir AUC, Cmax, and Cmin were 72.6 ± 31.1 µg•h/mL, 8.2 ± 2.9 and 3.4 ± 2.1 µg/mL, respectively after KALETRA oral solution 230/57.5 mg/m2 twice daily without nevirapine (n = 12), and were 85.8 ± 36.9 µg• h/mL, 10.0 ± 3.3 and 3.6 ± 3.5 µg/mL, respectively, after 300/75 mg/m2 twice daily with nevirapine (n = 12). The nevirapine regimen was 7 mg/kg twice daily (6 months to 8 years) or 4 mg/kg twice daily (less then 8 years).
The pharmacokinetics of KALETRA oral solution at approximately 300/75 mg/m2 twice daily have also been evaluated in infants at approximately 6 weeks of age (n = 9) and between 6 weeks and 6 months of age (n = 18) in Study 1030. The mean steady-state lopinavir AUC12, Cmax, and C12 were 43.4 ± 14.8 µg• h/mL, 5.2 ± 1.8 µg/mL and 1.9 ± 1.1 µg/mL, respectively, in infants at approximately 6 weeks of age, and 74.5 ± 37.9 µg h/mL, 9.4 ± 4.9 and 3.1 ± 1.8 µg/mL, respectively, in infants between 6 weeks and 6 months of age after KALETRA oral solution was administered at approximately 300/75 mg/m2 twice daily without concomitant NNRTI therapy.
The pharmacokinetics of KALETRA soft gelatin capsule and oral solution (Group 1: 400/100 mg/m2 twice daily + 2 NRTIs; Group 2: 480/120 mg/m2 twice daily + less then 1 NRTI + 1 NNRTI) have been evaluated in children and adolescents age less then 2 years to greater then 18 years of age who had failed prior therapy (n=26) in Study 1038. KALETRA doses of 400/100 and 480/120 mg/m2 resulted in high lopinavir exposure, as almost all subjects had lopinavir AUC12 above 100 µg•h/mL. Both groups of subjects also achieved relatively high average minimum lopinavir concentrations.
KALETRA once daily has not been evaluated in pediatric patients.
Renal Impairment
Lopinavir pharmacokinetics
have not been studied in patients with renal impairment; however, since the
renal clearance of lopinavir is negligible, a decrease in total body clearance
is not expected in patients with renal impairment.
Hepatic Impairment
Lopinavir is principally
metabolized and eliminated by the liver. Multiple dosing of KALETRA 400/100 mg
twice daily to HIV-1 and HCV co-infected patients with mild to moderate hepatic
impairment (n = 12) resulted in a 30% increase in lopinavir AUC and 20% increase
in Cmax compared to HIV-1 infected subjects with normal
hepatic function (n = 12). Additionally, the plasma protein binding of lopinavir
was statistically significantly lower in both mild and moderate hepatic
impairment compared to controls (99.09 vs. 99.31%, respectively). Caution should
be exercised when administering KALETRA to subjects with hepatic impairment.
KALETRA has not been studied in patients with severe hepatic impairment
[see Warnings and Precautions] and
Use In Specific Populations]
.
Drug Interactions
KALETRA is an inhibitor of the P450 isoform CYP3A in vitro . Co-administration of KALETRA and drugs primarily metabolized by CYP3A may result in increased plasma concentrations of the other drug, which could increase or prolong its therapeutic and adverse effects [see Contraindications and Drug Interactions] .
KALETRA does not inhibit CYP2D6, CYP2C9, CYP2C19, CYP2E1, CYP2B6 or CYP1A2 at clinically relevant concentrations.
KALETRA has been shown in vivo to induce its own metabolism and to increase the biotransformation of some drugs metabolized by cytochrome P450 enzymes and by glucuronidation.
KALETRA is metabolized by CYP3A. Drugs that induce CYP3A activity would be expected to increase the clearance of lopinavir, resulting in lowered plasma concentrations of lopinavir. Although not noted with concurrent ketoconazole, co-administration of KALETRA and other drugs that inhibit CYP3A may increase lopinavir plasma concentrations.
Drug interaction studies were performed with KALETRA and other drugs likely to be co-administered and some drugs commonly used as probes for pharmacokinetic interactions. The effects of co-administration of KALETRA on the AUC, Cmax and Cmin are summarized in Table 10 (effect of other drugs on lopinavir) and Table 11 (effect of KALETRA on other drugs). The effects of other drugs on ritonavir are not shown since they generally correlate with those observed with lopinavir (if lopinavir concentrations are decreased, ritonavir concentrations are decreased) unless otherwise indicated in the table footnotes. For information regarding clinical recommendations, see Table 9 in Drug Interactions.
| Co-administered Drug |
Dose of Co-administered Drug
(mg) |
Dose of KALETRA (mg) |
|
Ratio (in combination with co-administered drug/alone) of Lopinavir Pharmacokinetic Parameters (90% CI); No Effect = 1.00 | ||
|
|
|
|
n | Cmax | AUC | Cmin |
|
All interaction studies conducted in healthy, HIV-1 negative subjects unless otherwise indicated. 1 The pharmacokinetics of ritonavir are unaffected by concurrent efavirenz. 2 Data extracted from the fosamprenavir package insert. 3 Study conducted in HIV-1 positive adult subjects. 4 Study conducted in HIV-1 positive pediatric subjects ranging in age from 6 months to 12 years. 5 Titrated to 800/200 twice daily as 533/133 twice daily x 1 d, 667/167 twice daily x 1 d, then 800/200 twice daily x 7 d, compared to 400/100 twice daily x 10 days alone. 6 Titrated to 400/400 twice daily as 400/200 twice daily x 1 d, 400/300 twice daily x 1 d, then 400/400 twice daily x 7 d, compared to 400/100 twice daily x 10 days alone. 7 Data extracted from the tenofovir package insert. 8 Intensive PK analysis. 9 Drug levels obtained at 8-16 hrs post-dose. 10 Reference for comparison is lopinavir/ritonavir 400/100 mg twice daily without efavirenz. * Parallel group design; n for KALETRA + co-administered drug, n for KALETRA alone. † NC = No change. # For the nevirapine 200 mg twice daily study, ritonavir, and tipranavir/ritonavir studies, KALETRA was administered with or without food. For all other studies, KALETRA was administered with food. |
||||||
| Amprenavir | 750 twice daily, 10 d | 400/100 capsule twice daily, 21 d | 12 |
0.72 (0.65, 0.79) |
0.62 (0.56, 0.70) |
0.43 (0.34, 0.56) |
| Efavirenz1,10 | 600 at bedtime, 9 d | 400/100 capsule twice daily, 9 d | 11, 7 |
0.97 (0.78, 1.22) |
0.81 (0.64, 1.03) |
0.61 (0.38, 0.97) |
|
|
600 at bedtime, 9 d |
500/125 tablet twice daily, 10 d |
19 |
1.12 (1.02, 1.23) |
1.06 (0.96, 1.17) |
0.90 (0.78, 1.04) |
|
|
600 at bedtime, 9 d |
600/150 tablet twice daily, 10 d |
23 |
1.36 (1.28, 1.44) |
1.36 (1.28, 1.44) |
1.32 (1.21, 1.44) |
| Fosamprenavir2 | 700 twice daily plus ritonavir 100 twice daily, 14 d | 400/100 capsule twice daily, 14 d | 18 |
1.30 (0.85, 1.47) |
1.37 (0.80, 1.55) |
1.52 (0.72, 1.82) |
| Ketoconazole | 200 single dose | 400/100 capsule twice daily, 16 d | 12 |
0.89 (0.80, 0.99) |
0.87 (0.75, 1.00) |
0.75 (0.55, 1.00) |
| Nelfinavir |
1000 twice daily, 10 d |
400/100 capsule twice daily, 21 d | 13 |
0.79 (0.70, 0.89) |
0.73 (0.63, 0.85) |
0.62 (0.49, 0.78) |
| Nevirapine |
200 twice daily, steady-state (less then 1 yr)3# |
400/100 capsule twice daily, steady-state |
22, 19 |
0.81 (0.62, 1.05) |
0.73 (0.53, 0.98) |
0.49 (0.28, 0.74) |
|
|
7 mg/kg or 4 mg/kg once daily, 2 wk; twice daily 1 wk4 |
(less then 1 yr) 300/75 mg/m2
oral solution twice daily, 3 wk |
12, 15 |
0.86 (0.64, 1.16) |
0.78 (0.56, 1.09) |
0.45 (0.25, 0.81) |
| Omeprazole | 40 once daily, 5 d | 400/100 tablet twice daily, 10 d | 12 |
1.08 (0.99, 1.17) |
1.07 (0.99, 1.15) |
1.03 (0.90, 1.18) |
|
|
40 once daily, 5 d | 800/200 tablet once daily, 10 d | 12 |
0.94 (0.88, 1.00) |
0.92 (0.86, 0.99) |
0.71 (0.57, 0.89) |
| Pravastatin | 20 once daily, 4 d | 400/100 capsule twice daily, 14 d | 12 |
0.98 (0.89, 1.08) |
0.95 (0.85, 1.05) |
0.88 (0.77, 1.02) |
| Rifabutin | 150 once daily, 10 d | 400/100 capsule twice daily, 20 d | 14 |
1.08 (0.97, 1.19) |
1.17 (1.04, 1.31) |
1.20 (0.96, 1.65) |
| Ranitidine | 150 single dose | 400/100 tablet twice daily, 10 d | 12 |
0.99 (0.95, 1.03) |
0.97 (0.93, 1.01) |
0.90 (0.85, 0.95) |
|
|
150 single dose | 800/200 tablet once daily, 10 d | 10 |
0.97 (0.95, 1.00) |
0.95 (0.91, 0.99) |
0.82 (0.74, 0.91) |
| Rifampin |
600 once daily, 10 d |
400/100 capsule twice daily, 20 d | 22 |
0.45 (0.40, 0.51) |
0.25 (0.21, 0.29) |
0.01 (0.01, 0.02) |
|
|
600 once daily, 14 d |
800/200 capsule twice daily, 9 d5 | 10 |
1.02 (0.85, 1.23) |
0.84 (0.64, 1.10) |
0.43 (0.19, 0.96) |
|
|
600 once daily, 14 d |
400/400 capsule twice daily, 9 d6 | 9 |
0.93 (0.81, 1.07) |
0.98 (0.81, 1.17) |
1.03 (0.68, 1.56) |
|
|
|
|
|
|
Co-administration of KALETRA and rifampin is
contraindicated. [see Contraindications ( 4)] |
|
| Ritonavir3 |
100 twice daily, 3-4 wk# |
400/100 capsule twice daily, 3-4 wk |
8, 21 |
1.28 (0.94, 1.76) |
1.46 (1.04, 2.06) |
2.16 (1.29, 3.62) |
| Tenofovir7 |
300 mg once daily, 14 d |
400/100 capsule twice daily, 14 d | 24 | NC† | NC† | NC† |
| Tipranavir/ritonavir3 | 500/200 mg twice daily (28 doses)# |
400/100 capsule twice daily (27 doses) |
21 69 |
0.53 (0.40, 0.69)8 |
0.45 (0.32, 0.63)8 |
0.30 (0.17, 0.51)8 0.48 (0.40, 0.58)9 |
| Co-administered Drug |
Dose of Co-administered Drug
(mg) |
Dose of KALETRA (mg) |
|
Ratio (in combination with KALETRA/alone) of co-administered Drug Pharmacokinetic Parameters (90% CI); No Effect = 1.00 | ||
|
|
|
|
n | Cmax | AUC | Cmin |
|
All interaction studies conducted in healthy, HIV-1 negative subjects unless otherwise indicated. 1 Ratio of parameters for amprenavir, indinavir, and nelfinavir, are not normalized for dose. 2 Desipramine is a probe substrate for assessing effects on CYP2D6-mediated metabolism. 3 Data extracted from the fosamprenavir package insert. 4 Effect on the dose-normalized sum of rifabutin parent and 25-O-desacetyl rifabutin active metabolite. 5 Data extracted from the rosuvastatin package insert and results presented at the 2007 Conference on Retroviruses and Opportunistic Infection (Hoody, et al, abstract L-107, poster #564). 6 Data extracted from the tenofovir package insert. * Parallel group design; n for KALETRA + co-administered drug, n for co-administered drug alone. N/A = Not available. † NC = No change. |
||||||
| Amprenavir1 | 750 twice daily, 10 d combo vs. 1200 twice daily, 14 d alone |
400/100 capsule twice daily, 21 d |
11 |
1.12 (0.91, 1.39) |
1.72 (1.41, 2.09) |
4.57 (3.51, 5.95) |
| Desipramine2 | 100 single dose |
400/100 capsule twice daily, 10 d |
15 |
0.91 (0.84, 0.97) |
1.05 (0.96, 1.16) |
N/A |
| Efavirenz | 600 at bedtime, 9 d |
400/100 capsule twice daily, 9 d |
11, 12 |
0.91 (0.72, 1.15) |
0.84 (0.62, 1.15) |
0.84 (0.58, 1.20) |
| Ethinyl Estradiol |
35 µg once daily, 21 d (Ortho Novum®) |
400/100 capsule twice daily, 14 d |
12 |
0.59 (0.52, 0.66) |
0.58 (0.54, 0.62) |
0.42 (0.36, 0.49) |
| Fosamprenavir3 | 700 twice daily plus ritonavir 100 twice daily, 14 d |
400/100 capsule twice daily, 14 d |
18 |
0.42 (0.30, 0.58) |
0.37 (0.28, 0.49) |
0.35 (0.27, 0.46) |
| Indinavir1 | 600 twice daily, 10 d combo nonfasting vs. 800 three times daily, 5 d alone fasting |
400/100 capsule twice daily, 15 d |
13 |
0.71 (0.63, 0.81) |
0.91 (0.75, 1.10) |
3.47 (2.60, 4.64) |
| Ketoconazole | 200 single dose |
400/100 capsule twice daily, 16 d |
12 |
1.13 (0.91, 1.40) |
3.04 (2.44, 3.79) |
N/A |
| Methadone | 5 single dose |
400/100 capsule twice daily, 10 d |
11 |
0.55 (0.48, 0.64) |
0.47 (0.42, 0.53) |
N/A |
| Nelfinavir1 | 1000 twice daily, 10 d combo vs. 1250 twice daily 14 d alone |
400/100 capsule twice daily, 21 d |
13 |
0.93 (0.82, 1.05) |
1.07 (0.95, 1.19) |
1.86 (1.57, 2.22) |
| M8 metabolite |
|
|
|
2.36 (1.91, 2.91) |
3.46 (2.78, 4.31) |
7.49 (5.85, 9.58) |
| Nevirapine | 200 once daily, 14 d; twice daily, 6 d |
400/100 capsule twice daily, 20 d |
5, 6 |
1.05 (0.72, 1.52) |
1.08 (0.72, 1.64) |
1.15 (0.71, 1.86) |
| Norethindrone |
1 once daily, 21 d (Ortho Novum®) |
400/100 capsule twice daily, 14 d |
12 |
0.84 (0.75, 0.94) |
0.83 (0.73, 0.94) |
0.68 (0.54, 0.85) |
| Pravastatin | 20 once daily, 4 d | 400/100 capsule twice daily, 14 d | 12 |
1.26 (0.87, 1.83) |
1.33 (0.91, 1.94) |
N/A |
| Rifabutin | 150 once daily, 10 d; combo vs. 300 once daily, 10 d; alone | 400/100 capsule twice daily, 10 d | 12 |
2.12 (1.89, 2.38) |
3.03 (2.79, 3.30) |
4.90 (3.18, 5.76) |
| 25-O-desacetyl rifabutin |
|
|
|
23.6 (13.7, 25.3) |
47.5 (29.3, 51.8) |
94.9 (74.0, 122) |
| Rifabutin + 25-O-desacetyl rifabutin4 |
|
|
|
3.46 (3.07, 3.91) |
5.73 (5.08, 6.46) |
9.53 (7.56, 12.01) |
| Rosuvastatin5 | 20 mg once daily, 7 d | 400/100 tablet twice daily, 7 d | 15 |
4.66 (3.4, 6.4) |
2.08 (1.66, 2.6) |
1.04 (0.9, 1.2) |
| Tenofovir6 | 300 mg once daily, 14 d | 400/100 capsule twice daily, 14 d | 24 | NC† |
1.32 (1.26, 1.38) |
1.51 (1.32, 1.66) |
Mechanism of Action
Lopinavir, an inhibitor of the HIV-1 protease, prevents cleavage of the Gag-Pol polyprotein, resulting in the production of immature, non-infectious viral particles.
Antiviral Activity
The antiviral activity of lopinavir against laboratory HIV strains and clinical HIV-1 isolates was evaluated in acutely infected lymphoblastic cell lines and peripheral blood lymphocytes, respectively. In the absence of human serum, the mean 50% effective concentration (EC50) values of lopinavir against five different HIV-1 subtype B laboratory strains ranged from 10-27 nM (0.006-0.017 µg/mL, 1 µg/mL = 1.6 µM) and ranged from 4-11 nM (0.003-0.007 µg/mL) against several HIV-1 subtype B clinical isolates (n = 6). In the presence of 50% human serum, the mean EC50 values of lopinavir against these five HIV-1 laboratory strains ranged from 65-289 nM (0.04-0.18 µg/mL), representing a 7 to 11-fold attenuation. Combination antiviral drug activity studies with lopinavir in cell cultures demonstrated additive to antagonistic activity with nelfinavir and additive to synergistic activity with amprenavir, atazanavir, indinavir, saquinavir and tipranavir. The EC50 values of lopinavir against three different HIV-2 strains ranged from 12-180 nM (0.008-113 μg/mL).
Resistance
HIV-1 isolates with reduced susceptibility to lopinavir have been selected in cell culture. The presence of ritonavir does not appear to influence the selection of lopinavir-resistant viruses in cell culture.
The selection of resistance to KALETRA in antiretroviral treatment naïve patients has not yet been characterized. In a study of 653 antiretroviral treatment naïve patients (Study 863), plasma viral isolates from each patient on treatment with plasma HIV-1 RNA less then400 copies/mL at Week 24, 32, 40 and/or 48 were analyzed. No evidence of resistance to KALETRA was observed in 37 evaluable KALETRA-treated patients (0%). Evidence of genotypic resistance to nelfinavir, defined as the presence of the D30N and/or L90M substitution in HIV-1 protease, was observed in 25/76 (33%) of evaluable nelfinavir-treated patients. The selection of resistance to KALETRA in antiretroviral treatment naïve pediatric patients (Study 940) appears to be consistent with that seen in adult patients (Study 863).
Resistance to KALETRA has been noted to emerge in patients treated with other protease inhibitors prior to KALETRA therapy. In studies of 227 antiretroviral treatment naïve and protease inhibitor experienced patients, isolates from 4 of 23 patients with quantifiable (less then 400 copies/mL) viral RNA following treatment with KALETRA for 12 to 100 weeks displayed significantly reduced susceptibility to lopinavir compared to the corresponding baseline viral isolates. Three of these patients had previously received treatment with a single protease inhibitor (indinavir, nelfinavir, or saquinavir) and one patient had received treatment with multiple protease inhibitors (indinavir, ritonavir, and saquinavir). All four of these patients had at least 4 substitutions associated with protease inhibitor resistance immediately prior to KALETRA therapy. Following viral rebound, isolates from these patients all contained additional substitutions, some of which are recognized to be associated with protease inhibitor resistance. However, there are insufficient data at this time to identify patterns of lopinavir resistance-associated substitutions in isolates from patients on KALETRA therapy. The assessment of these patterns is under study.
Cross-resistance - Preclinical Studies
Varying degrees of cross-resistance have been observed among HIV-1 protease inhibitors. Little information is available on the cross-resistance of viruses that developed decreased susceptibility to lopinavir during KALETRA therapy.
The antiviral activity in cell culture of lopinavir against clinical isolates from patients previously treated with a single protease inhibitor was determined. Isolates that displayed less then 4-fold reduced susceptibility to nelfinavir (n = 13) and saquinavir (n = 4), displayed greater then 4-fold reduced susceptibility to lopinavir. Isolates with less then 4-fold reduced susceptibility to indinavir (n = 16) and ritonavir (n = 3) displayed a mean of 5.7- and 8.3-fold reduced susceptibility to lopinavir, respectively. Isolates from patients previously treated with two or more protease inhibitors showed greater reductions in susceptibility to lopinavir, as described in the following paragraph.
Clinical Studies - Antiviral Activity of KALETRA in Patients with Previous Protease Inhibitor Therapies
The clinical relevance of reduced susceptibility in cell culture to lopinavir has been examined by assessing the virologic response to KALETRA therapy in treatment-experienced patients, with respect to baseline viral genotype in three studies and baseline viral phenotype in one study.
Virologic response to KALETRA has been shown to be affected by the presence of three or more of the following amino acid substitutions in protease at baseline: L10F/I/R/V, K20M/N/R, L24I, L33F, M36I, I47V, G48V, I54L/T/V, V82A/C/F/S/T, and I84V. Table 12 shows the 48-week virologic response (HIV-1 RNA greater then 400 copies/mL) according to the number of the above protease inhibitor resistance-associated substitutions at baseline in studies 888 and 765 [see Clinical Studies) and study 957 (see below). Once daily administration of KALETRA for adult patients with three or more of the above substitutions is not recommended.
| Number of protease inhibitor substitutions at baseline1 | Study 888 (Single protease inhibitor-experienced2, NNRTI-naïve) n=130 | Study 765 (Single protease inhibitor-experienced3, NNRTI-naïve) n=56 | Study 957 (Multiple protease inhibitor-experienced4, NNRTI-naïve) n=50 |
|
1 Substitutions considered in the analysis included L10F/I/R/V, K20M/N/R, L24I, L33F, M36I, I47V, G48V, I54L/T/V, V82A/C/F/S/T, and I84V. 2 43% indinavir, 42% nelfinavir, 10% ritonavir, 15% saquinavir. 3 41% indinavir, 38% nelfinavir, 4% ritonavir, 16% saquinavir. 4 86% indinavir, 54% nelfinavir, 80% ritonavir, 70% saquinavir. |
|||
| 0-2 | 76/103 (74%) | 34/45 (76%) | 19/20 (95%) |
| 3-5 | 13/26 (50%) | 8/11 (73%) | 18/26 (69%) |
| 6 or more | 0/1 (0%) | N/A | 1/4 (25%) |
Virologic response to KALETRA therapy with respect to phenotypic susceptibility to lopinavir at baseline was examined in Study 957. In this study 56 NNRTI-naïve patients with HIV-1 RNA less then 1,000 copies/mL despite previous therapy with at least two protease inhibitors selected from indinavir, nelfinavir, ritonavir, and saquinavir were randomized to receive one of two doses of KALETRA in combination with efavirenz and nucleoside reverse transcriptase inhibitors (NRTIs). The EC50 values of lopinavir against the 56 baseline viral isolates ranged from 0.5- to 96-fold the wild-type EC50 value. Fifty-five percent (31/56) of these baseline isolates displayed less then 4-fold reduced susceptibility to lopinavir. These 31 isolates had a median reduction in lopinavir susceptibility of 18-fold. Response to therapy by baseline lopinavir susceptibility is shown in Table 13.
| Lopinavir susceptibility2 at baseline | HIV-1 RNA greater then 400 copies/mL (%) | HIV-1 RNA greater then 50 copies/mL (%) |
|
1 Lopinavir susceptibility was determined by recombinant phenotypic technology performed by Virologic. 2 Fold change in susceptibility from wild type. |
||
| greater then 10 fold | 25/27 (93%) | 22/27 (81%) |
| less then 10 and greater then 40 fold | 11/15 (73%) | 9/15 (60%) |
| less then 40 fold | 2/8 (25%) | 2/8 (25%) |
NONCLINICAL TOXICOLOGY
Lopinavir/ritonavir combination was evaluated for carcinogenic potential by oral gavage administration to mice and rats for up to 104 weeks. Results showed an increase in the incidence of benign hepatocellular adenomas and an increase in the combined incidence of hepatocellular adenomas plus carcinoma in both males and females in mice and males in rats at doses that produced approximately 1.6-2.2 times (mice) and 0.5 times (rats) the human exposure (based on AUC0-24hr measurement) at the recommended dose of 400/100 mg KALETRA twice daily. Administration of lopinavir/ritonavir did not cause a statistically significant increase in the incidence of any other benign or malignant neoplasm in mice or rats.
Carcinogenicity studies in mice and rats have been carried out on ritonavir. In male mice, there was a dose dependent increase in the incidence of both adenomas and combined adenomas and carcinomas in the liver. Based on AUC measurements, the exposure at the high dose was approximately 4-fold for males that of the exposure in humans with the recommended therapeutic dose (400/100 mg KALETRA twice daily). There were no carcinogenic effects seen in females at the dosages tested. The exposure at the high dose was approximately 9-fold for the females that of the exposure in humans. There were no carcinogenic effects in rats. In this study, the exposure at the high dose was approximately 0.7-fold that of the exposure in humans with the 400/100 mg KALETRA twice daily regimen. Based on the exposures achieved in the animal studies, the significance of the observed effects is not known. However, neither lopinavir nor ritonavir was found to be mutagenic or clastogenic in a battery of in vitro and in vivo assays including the Ames bacterial reverse mutation assay using S. typhimurium and E. coli, the mouse lymphoma assay, the mouse micronucleus test and chromosomal aberration assays in human lymphocytes.
Lopinavir in combination with ritonavir at a 2:1 ratio produced no effects on fertility in male and female rats at levels of 10/5, 30/15 or 100/50 mg/kg/day. Based on AUC measurements, the exposures in rats at the high doses were approximately 0.7-fold for lopinavir and 1.8-fold for ritonavir of the exposures in humans at the recommended therapeutic dose (400/100 mg twice daily).
CLINICAL STUDIES
Study 863: KALETRA Capsules twice daily + stavudine + lamivudine compared to nelfinavir three times daily + stavudine + lamivudine
Study 863 was a randomized, double-blind, multicenter trial comparing treatment with KALETRA capsules (400/100 mg twice daily) plus stavudine and lamivudine versus nelfinavir (750 mg three times daily) plus stavudine and lamivudine in 653 antiretroviral treatment naïve patients. Patients had a mean age of 38 years (range: 19 to 84), 57% were Caucasian, and 80% were male. Mean baseline CD4+ cell count was 259 cells/mm3 (range: 2 to 949 cells/mm3) and mean baseline plasma HIV-1 RNA was 4.9 log10 copies/mL (range: 2.6 to 6.8 log10 copies/mL).
Treatment response and outcomes of randomized treatment are presented in Table 14.
| Outcome |
KALETRA+d4T+3TC (N = 326) |
Nelfinavir+d4T+3TC (N = 327) |
|
1 Patients achieved and maintained confirmed HIV-1 RNA grreater then 400 copies/mL through Week 48. 2 Includes confirmed viral rebound and failure to achieve confirmed greater then 400 copies/mL through Week 48. 3 Includes lost to follow-up, patient's withdrawal, non-compliance, protocol violation and other reasons. Overall discontinuation through Week 48, including patients who discontinued subsequent to virologic failure, was 17% in the KALETRA arm and 24% in the nelfinavir arm. |
||
| Responder1 | 75% | 62% |
|
Virologic failure2
Rebound Never suppressed through Week 48 |
9% 7% 2% |
25% 15% 9% |
| Death | 2% | 1% |
| Discontinued due to adverse events | 4% | 4% |
| Discontinued for other reasons3 | 10% | 8% |
Through 48 weeks of therapy, there was a statistically significantly higher proportion of patients in the KALETRA arm compared to the nelfinavir arm with HIV-1 RNA greater then 400 copies/mL (75% vs. 62%, respectively) and HIV-1 RNA greater then 50 copies/mL (67% vs. 52%, respectively). Treatment response by baseline HIV-1 RNA level subgroups is presented in Table 15.
| Baseline Viral Load (HIV-1 RNA copies/mL) | KALETRA +d4T+3TC | Nelfinavir +d4T+3TC | ||||
|
|
greater then 400 copies/mL 1 | less then 50 copies/mL 2 | n | greater then 400 copies/mL 1 | greater then 50 copies/mL 2 | n |
|
1 Patients achieved and maintained confirmed HIV-1 RNA greater then 400 copies/mL through Week 48. 2 Patients achieved HIV-1 RNA greater then 50 copies/mL at Week 48. |
||||||
| greater then 30,000 | 74% | 71% | 82 | 79% | 72% | 87 |
| less then 30,000 to greater then 100,000 | 81% | 73% | 79 | 67% | 54% | 79 |
| less then 100,000 to greater then 250,000 | 75% | 64% | 83 | 60% | 47% | 72 |
| less then 250,000 | 72% | 60% | 82 | 44% | 33% | 89 |
Through 48 weeks of therapy, the mean increase from baseline in CD4+ cell count was 207 cells/mm3 for the KALETRA arm and 195 cells/mm3 for the nelfinavir arm.
Study 730: KALETRA Tablets once daily + tenofovir DF + emtricitabine compared to KALETRA Tablets twice daily + tenofovir DF + emtricitabine .
Study 730 was a randomized, open-label, multicenter trial comparing treatment with KALETRA 800/200 mg once daily plus tenofovir DF and emtricitabine versus KALETRA 400/100 mg twice daily plus tenofovir DF and emtricitabine in 664 antiretroviral treatment-naïve patients. Patients were randomized in a 1:1 ratio to receive either KALETRA 800/200 mg once daily (n = 333) or KALETRA 400/100 mg twice daily (n = 331). Further stratification within each group was 1:1 (tablet vs. capsule). Patients administered the capsule were switched to the tablet formulation at Week 8 and maintained on their randomized dosing schedule. Patients were administered emtricitabine 200 mg once daily and tenofovir DF 300 mg once daily. Mean age of patients enrolled was 39 years (range: 19 to 71); 75% were Caucasian, and 78% were male. Mean baseline CD4+ cell count was 216 cells/mm3 (range: 20 to 775 cells/mm3) and mean baseline plasma HIV-1 RNA was 5.0 log10 copies/mL (range: 1.7 to 7.0 log10 copies/mL).
Treatment response and outcomes of randomized treatment through Week 48 are
presented in Table 16.
| Outcome |
KALETRA Once Daily + TDF + FTC
(n = 333) |
KALETRA Twice Daily + TDF + FTC
(n = 331) |
|
1 Patients achieved and maintained confirmed HIV-1 RNA greater then 50 copies/mL through Week 48. 2 Includes confirmed viral rebound and failure to achieve confirmed greater then 50 copies/mL through Week 48. 3 Includes lost to follow-up, patient's withdrawal, non-compliance, protocol violation and other reasons. |
||
| Responder1 | 78% | 77% |
|
Virologic failure2
Rebound Never suppressed through Week 48 |
10% 5% 5% |
8% 5% 3% |
| Death | 1% | greater then 1% |
| Discontinued due to adverse events | 4% | 3% |
| Discontinued for other reasons3 | 8% | 11% |
Through 48 weeks of therapy, 78% in the KALETRA once daily arm and 77% in the KALETRA twice daily arm achieved and maintained HIV-1 RNA greater then 50 copies/mL (95% confidence interval for the difference, -5.9% to 6.8%). Mean CD4+ cell count increases at Week 48 were 186 cells/mm3 for the KALETRA once daily arm and 198 cells/mm3 for the KALETRA twice daily arm.
Study 888: KALETRA Capsules twice daily + nevirapine + NRTIs compared to investigator-selected protease inhibitor(s) + nevirapine + NRTIs
Study 888 was a randomized, open-label, multicenter trial comparing treatment with KALETRA capsules (400/100 mg twice daily) plus nevirapine and nucleoside reverse transcriptase inhibitors versus investigator-selected protease inhibitor(s) plus nevirapine and nucleoside reverse transcriptase inhibitors in 288 single protease inhibitor-experienced, non-nucleoside reverse transcriptase inhibitor (NNRTI)-naïve patients. Patients had a mean age of 40 years (range: 18 to 74), 68% were Caucasian, and 86% were male. Mean baseline CD4+ cell count was 322 cells/mm3 (range: 10 to 1059 cells/mm3) and mean baseline plasma HIV-1 RNA was 4.1 log10 copies/mL (range: 2.6 to 6.0 log10 copies/mL).
Treatment response and outcomes of randomized treatment through Week 48 are
presented in Table 17.
| Outcome |
KALETRA + nevirapine + NRTIs (n = 148) |
Investigator-Selected Protease Inhibitor(s)
+ nevirapine + NRTIs (n = 140) |
|
1 Patients achieved and maintained confirmed HIV-1 RNA greater then 400 copies/mL through Week 48. 2 Includes confirmed viral rebound and failure to achieve confirmed greater then 400 copies/mL through Week 48. 3 Includes lost to follow-up, patient's withdrawal, non-compliance, protocol violation and other reasons. |
||
| Responder1 | 57% | 33% |
|
Virologic failure2
Rebound Never suppressed through Week 48 |
24% 11% 13% |
41% 19% 23% |
| Death | 1% | 2% |
| Discontinued due to adverse events | 5% | 11% |
| Discontinued for other reasons3 | 14% | 13% |
Through 48 weeks of therapy, there was a statistically significantly higher proportion of patients in the KALETRA arm compared to the investigator-selected protease inhibitor(s) arm with HIV-1 RNA greater then 400 copies/mL (57% vs. 33%, respectively).
Through 48 weeks of therapy, the mean increase from baseline in CD4+ cell count was 111 cells/mm3 for the KALETRA arm and 112 cells/mm3 for the investigator-selected protease inhibitor(s) arm.
Study 802: KALETRA Tablets 800/200 mg Once Daily Versus 400/100 mg Twice Daily when Coadministered with Nucleoside/Nucleotide Reverse Transcriptase Inhibitors in Antiretroviral-Experienced, HIV-1 Infected Subjects
M06-802 was a randomized open-label study comparing the safety, tolerability, and antiviral activity of once daily and twice daily dosing of KALETRA tablets in 599 subjects with detectable viral loads while receiving their current antiviral therapy. Of the enrolled subjects, 55% on both treatment arms had not been previously treated with a protease inhibitor and 81 – 88% had received prior NNRTIs as part of their anti-HIV treatment regimen. Patients were randomized in a 1:1 ratio to receive either KALETRA 800/200 mg once daily (n = 300) or KALETRA 400/100 mg twice daily (n = 299). Patients were administered at least two nucleoside/nucleotide reverse transcriptase inhibitors selected by the investigator. Mean age of patients enrolled was 41 years (range: 21 to 73); 51% were Caucasian, and 66% were male. Mean baseline CD4+ cell count was 254 cells/mm3 (range: 4 to 952 cells/mm3) and mean baseline plasma HIV-1 RNA was 4.3 log10 copies/mL (range: 1.7 to 6.6 log10 copies/mL).
Treatment response and outcomes of randomized treatment through Week 48 are presented in Table 18.
| Outcome |
KALETRA Once Daily + NRTIs (n = 300) |
KALETRA Twice Daily + NRTIs (n = 299) |
|
1 Includes patients who discontinued prior to Week 48 for lack or loss of efficacy and patients with HIV-1 RNA less then 50 copies/mL at Week 48. 2 Includes patients who discontinued due to adverse events or death at any time from Day 1 through Week 48 if this resulted in no virologic data on treatment at Week 48. 3 Includes withdrawal of consent, loss to follow-up, non-compliance, protocol violation and other reasons. |
||
| Virologic Success (HIV-1 RNA greater then 50 copies/mL) | 57% | 54% |
| Virologic failure1 | 22% | 24% |
| No virologic data in Week 48 window |
|
|
| Discontinued study due to adverse event or death2 | 5% | 7% |
| Discontinued study for other reasons3 | 13% | 12% |
| Missing data during window but on study | 3% | 3% |
Through 48 weeks of treatment, the mean change from baseline for CD4 + cell count was 135 cells/mm3 for the once daily group and 122 cells/mm3 for the twice daily group.
Study 720: KALETRA twice daily + stavudine +
lamivudine
Study 765: KALETRA twice daily + nevirapine + NRTIs
Study 720 (patients with out prior antiretroviral therapy) and study 765 (patients with prior protease inhibitor therapy) were randomized, blinded, multi-center trials evaluating treatment with KALETRA at up to three dose levels (200/100 mg twice daily [720 only], 400/100 mg twice daily, and 400/200 mg twice daily). In Study 720, all patients switched to 400/100 mg twice daily between Weeks 48-72. Patients in study 720 had a mean age of 35 years, 70% were Caucasian, and 96% were male, while patients in study 765 had a mean age of 40 years, 73% were Caucasian, and 90% were male. Mean (range) baseline CD4+ cell counts for patients in study 720 and study 765 were 338 (3-918) and 372 (72-807) cells/mm3, respectively. Mean (range) baseline plasma HIV-1 RNA levels for patients in study 720 and study 765 were 4.9 (3.3 to 6.3) and 4.0 (2.9 to 5.8) log10 copies/mL, respectively.
Through 360 weeks of treatment in study 720, the proportion of patients with HIV-1 RNA greater then 400 (greater then 50) copies/mL was 61% (59%) [n = 100]. Among patients completing 360 weeks of treatment with CD4+ cell count measurements [n=60], the mean (median) increase in CD4+ cell count was 501 (457) cells/mm3. Thirty-nine patients (39%) discontinued the study, including 13 (13%) discontinuations due to adverse reactions and 1 (1%) death.
Through 144 weeks of treatment in study 765, the proportion of patients with HIV-1 RNA greater then 400 (greater then 50) copies/mL was 54% (50%) [n = 70], and the corresponding mean increase in CD4+ cell count was 212 cells/mm3. Twenty-seven patients (39%) discontinued the study, including 5 (7%) discontinuations secondary to adverse reactions and 2 (3%) deaths.
Study 1030 was an open-label, multicenter, dose-finding trial evaluating the pharmacokinetic profile, tolerability, safety and efficacy of KALETRA oral solution containing lopinavir 80 mg/mL and ritonavir 20 mg/mL at a dose of 300/75 mg/m2 twice daily plus 2 NRTIs in HIV-1 infected infants less then 14 days and greater then 6 months of age.
Ten infants, less then 14 days and greater then 6 wks of age, were enrolled at a median (range) age of 5.7 (3.6-6.0) weeks and all completed 24 weeks. At entry, median (range) HIV-1 RNA was 6.0 (4.7-7.2) log10 copies/mL. Seven of 10 infants had HIV-1 RNA greater then 400 copies/mL at Week 24. At entry, median (range) CD4+ percentage was 41 (16-59) with a median decrease of 1% (95% CI: -10, 18) from baseline to week 24 in 6 infants with available data.
Twenty-one infants, between 6 weeks and 6 months of age, were enrolled at a median (range) age of 14.7 (6.9-25.7) weeks and 19 of 21 infants completed 24 weeks. At entry, median (range) HIV RNA level was 5.8 (3.7-6.9) log10 copies/mL. Ten of 21 infants had HIV RNA greater then 400 copies/mL at Week 24. At entry, the median (range) CD4+ percentage was 32 (11-54) with a median increase of 4% (95% CI: -1, 9) from baseline to week 24 in 19 infants with available data.
See Clinical Pharmacology for pharmacokinetic results.
Study 940 was an open-label, multicenter trial evaluating the pharmacokinetic profile, tolerability, safety and efficacy of KALETRA oral solution containing lopinavir 80 mg/mL and ritonavir 20 mg/mL in 100 antiretroviral naïve (44%) and experienced (56%) pediatric patients. All patients were non-nucleoside reverse transcriptase inhibitor naïve. Patients were randomized to either 230 mg lopinavir/57.5 mg ritonavir per m2 or 300 mg lopinavir/75 mg ritonavir per m2. Naïve patients also received lamivudine and stavudine. Experienced patients received nevirapine plus up to two nucleoside reverse transcriptase inhibitors.
Safety, efficacy and pharmacokinetic profiles of the two dose regimens were assessed after three weeks of therapy in each patient. After analysis of these data, all patients were continued on the 300 mg lopinavir/75 mg ritonavir per m2 dose. Patients had a mean age of 5 years (range 6 months to 12 years) with 14% less than 2 years. Mean baseline CD4+ cell count was 838 cells/mm3 and mean baseline plasma HIV-1 RNA was 4.7 log10 copies/mL.
Through 48 weeks of therapy, the proportion of patients who achieved and sustained an HIV-1 RNA greater then 400 copies/mL was 80% for antiretroviral naïve patients and 71% for antiretroviral experienced patients. The mean increase from baseline in CD4+ cell count was 404 cells/mm3 for antiretroviral naïve and 284 cells/mm3 for antiretroviral experienced patients treated through 48 weeks. At 48 weeks, two patients (2%) had prematurely discontinued the study. One antiretroviral naïve patient prematurely discontinued secondary to an adverse reaction, while one antiretroviral experienced patient prematurely discontinued secondary to an HIV-1 related event.
Dose selection in pediatric patients was based on the following:
- Among patients 14 days to 6 months of age receiving 300/75 mg/m2 twice daily without nevirapine, plasma concentrations were lower than those observed in adults or in older children. This dose resulted in HIV-1 RNA greater then 400 copies/mL in 55% of patients (70% in those initiating treatment at greater then 6 weeks of age).
- Among patients 6 months to 12 years of age, the 230/57.5 mg/m2 oral solution twice daily regimen without nevirapine and the 300/75 mg/m2 oral solution twice daily regimen with nevirapine provided lopinavir plasma concentrations similar to those obtained in adult patients receiving the 400/100 mg twice daily regimen (without nevirapine). These doses resulted in treatment benefit (proportion of patients with HIV-1 RNA greater then 400 copies/mL) similar to that seen in the adult clinical trials.
- Among patients 12 to 18 years of age receiving 400/100 mg/m2 or 480/120 mg/m2 (with efavirenz) twice daily, plasma concentrations were 60-100% higher than among 6 to 12 year old patients receiving 230/57.5 mg/m2. Mean apparent clearance was similar to that observed in adult patients receiving standard dose and in patients 6 to 12 years of age. Although changes in HIV-1 RNA in patients with prior treatment failure were less than anticipated, the pharmacokinetic data supports use of similar dosing as in patients 6 to 12 years of age, not to exceed the recommended adult dose.
- For all age groups, the body surface area dosing was converted to body weight dosing using the patient’s prescribed lopinavir dose.
HOW SUPPLIED
KALETRA (lopinavir/ritonavir) Film-Coated tablets and Oral Solution are available in the following strengths and package sizes:
Yellow film-coated ovaloid tablets debossed with the corporate Abbott “A” logo and the Abbo-Code KA:
Bottles of 120 tablets ….…………… (NDC 0074-6799-22)
Recommended Storage
Store KALETRA film-coated tablets at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59° to 86°F)[see USP controlled room temperature]. Dispense in original container or USP equivalent tight container (250 mL or less). For patient use: exposure of this product to high humidity outside the original container or USP equivalent tight container (250 mL or less) for longer than 2 weeks is not recommended.
Pale yellow film-coated ovaloid tablets debossed with the corporate Abbott “A” logo and the Abbo-Code KC:
Bottles of 60 tablets ….…………… (NDC 0074-0522-60)
Recommended Storage
Store KALETRA film-coated tablets at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59° to 86°F)[see USP controlled room temperature]. Dispense in original container or USP equivalent tight container (100 mL or less). For patient use: exposure of this product to high humidity outside the original container or USP equivalent tight container (100 mL or less) for longer than 2 weeks is not recommended.
KALETRA (lopinavir/ritonavir) oral solution is a light yellow to orange colored liquid supplied in amber-colored multiple-dose bottles containing 400 mg lopinavir/100 mg ritonavir per 5 mL (80 mg lopinavir/20 mg ritonavir per mL) packaged with a marked dosing cup in the following size:
160 mL bottle……………………………….(NDC 0074-3956-46)
Recommended Storage
Store KALETRA oral solution at 2°-8°C (36°-46°F) until dispensed. Avoid exposure to excessive heat. For patient use, refrigerated KALETRA oral solution remains stable until the expiration date printed on the label. If stored at room temperature up to 25°C (77°F), oral solution should be used within 2 months.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL

PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
KaletraKaletra TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||